Sep 25, 2025
From Bone to Flow: Comprehensive Flow Cytometry Protocol for Profiling B Cell Populations in Mouse Bone Marrow
- Cinthia Mariel Olexen1,
- Gisela María Suárez Formigo1,
- Tamara Menendez Medina1,
- Zaima Mazorra Herrera1
- 1Abu Dhabi Stem Cell Center, Abu Dhabi, United Arab Emirates

Protocol Citation: Cinthia Mariel Olexen, Gisela María Suárez Formigo, Tamara Menendez Medina, Zaima Mazorra Herrera 2025. From Bone to Flow: Comprehensive Flow Cytometry Protocol for Profiling B Cell Populations in Mouse Bone Marrow. protocols.io https://dx.doi.org/10.17504/protocols.io.e6nvw4o67lmk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 25, 2025
Last Modified: September 25, 2025
Protocol Integer ID: 228138
Keywords: Murine Bone marrow, B cells, Flow Cytometry, comprehensive flow cytometry protocol, flow cytometry, differentiated plasma cell, murine bone marrow, insights into memory cell formation, distinct expression patterns of cd43, understanding of immune regulation, diverse roles in immune function, bone marrow, memory cell formation, plasma cell, igd expression during the maturation process, immune function, immune regulation, cd43 expression, specific surface marker, b220
Abstract
B cell subsets in murine bone marrow play diverse roles in immune function and disease, making their identification and characterization essential. Flow cytometry enables effective discrimination between these subsets using specific surface markers. This protocol describes the use of key markers—B220, CD45, CD19, IgD, IgM, and CD43—to identify and analyze B cell populations.
B220, a pan-B cell marker, plays a critical role in distinguishing between the various maturation stages of B cells. It is particularly useful for tracking the transition of B cells from precursors to mature forms (Bleesing & Fleisher, 2002). CD45, another significant marker, is used to differentiate stages within the B cell lineage, providing insights into memory cell formation in mice (Bleesing & Fleisher, 2002). CD19 is a consistent marker expressed across different B cell stages and is essential for initiating signal transduction, which is crucial for B cell activation and development (Sato et al., 2004).
Accurate identification of B cells in the bone marrow requires the use of B cell-specific molecules, CD19 and/or B220, as surface markers. B220 and CD19 are present on the surface of all later-stage B lineage cells, except for a subpopulation of terminally differentiated plasma cells. To further differentiate B cell developmental stages, pre-pro B cells, pro-B cells, and pre-B cells are defined based on their distinct expression patterns of CD43. IgD and IgM are pivotal in identifying naive and early B cell stages, where IgM is generally expressed initially, followed by IgD expression during the maturation process. Their combined use facilitates the identification of transitional stages within the B cell lineage (Sato et al. 2004). Finally, CD43 expression is notably valuable for distinguishing B-1 cells, which differ from conventional B cells by being predominantly expressed in distinct subsets, such as those in the spleen (Wells et al., 1994).
This method provides a comprehensive approach to delineate B cell subset dynamics, summarizing and streamlining the experimental steps and data interpretation to facilitate its application across different studies, contributing significantly to the understanding of immune regulation and potential alterations leading to disease.
Attachments

Figure 2.pdf
125KB
Materials
Materials and reagents
Scissors
Tweezers
Dissection pins
70% ethanol
Pipette tips (10ul - 200ul - 1000ul)
Serological pipettes 5ml.
Sterile containers
18 G syringe needle
0.22 μm syringe filter
96-well plates (U-bottom)
Biohazard bag
1.5 mL microcentrifuge tubes
70 μm cell strainer.
Bovine serum albumin (BSA) (Sigma, catalog number: BP1600)
Mice (C57BL/6)
Gibco. Trypan Blue Solution, 0.4% (100 mL), Cat# 15250061
LIVE/DEAD Fixable Aqua Dead Cell Stain Kit, Cat# L34957
Rat anti-mouse monoclonal, CD19 (clone 6D5, BioLegend, APC-Cy7, Cat# 115530)
Rat anti-mouse monoclonal, CD43 (clone S7, BioLegend, AF700, Cat# 143214)
Rat anti-mouse monoclonal, IgM (clone RMM-1, BioLegend, PerCP-Cy7, Cat# 406514)
Rat anti-mouse monoclonal, B220/CD45R (clone RA3-6B2, BioLegend, FITC, Cat# 103206)
Rat anti-mouse monoclonal, CD45 (clone 30-F11, Invitrogen, PE-Texas Red, Cat# MCD4517)
Rat anti-mouse monoclonal, IgD (clone 11-26c, Invitrogen, PE, Cat# 12-5993-82)
Solutions:
PBS: Dulbecco’s PBS, pH 7.4
FACS buffer: PBS1x and BSA 1%
RPMI 1640 supplemented: RPMI 1640 supplemented with 10% (v/v) Fetal bovine serum and 1% (v/v) Penicillin/ Streptomycin (sRPMI).
Equipment:
Vortex
Centrifuge (bench-top, 15/50 mL tubes)
Centrifuge for microplates (plate centrifuge)
Pipettes (Eppendorf)
Serological pipette controller
Biosafety cabinet, Class II, Type A2
Flow cytometer (DxFlex BeckmanCoulter)
Software:
FlowJo software (FlowJo, LLC.)
Troubleshooting
Organ collection
Bone Marrow collection
Euthanize the mice and isolate the femur: Euthanize C57BL/6 mice under standard protocol or to the specifications of the users’ mouse facility.
Begin by spraying the entire mouse with 70% ethanol to ensure sterility and prevent infection.
Make a precise incision along the hind leg, starting at the foot and extending upward to the groin area, ensuring a clean cut through the skin.
Gently retract the skin towards the back, taking care to make an additional cut, if necessary, to fully expose the underlying muscle tissue.
Using fine scissors, meticulously dissect and remove the muscle tissue from the area between the knee joint.
Repeat with second hind leg to isolate two femurs in total.
Discard mouse carcass according to guidelines Institutional Animal Care.
Place femurs in an appropriate container with sRPMI and keep refrigerated at 4 °C until processing.
Cell isolation
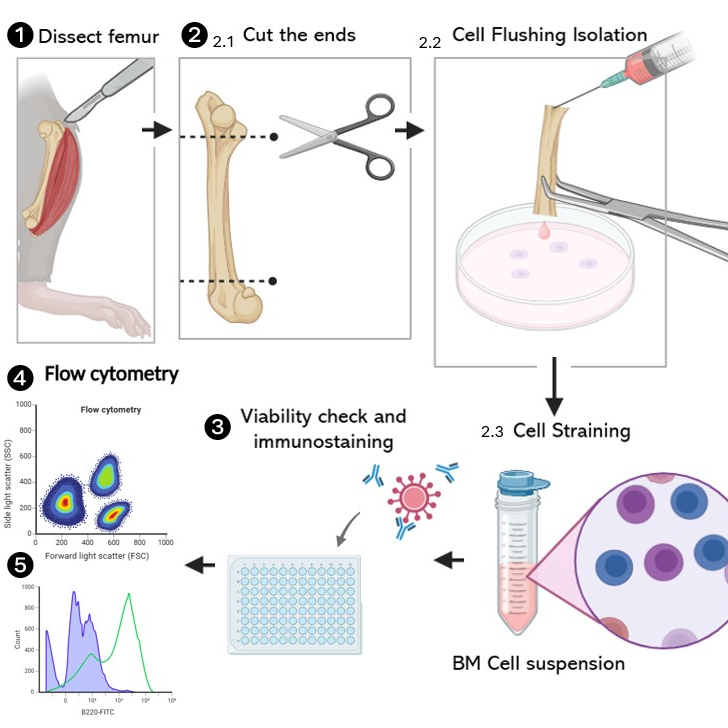
Bone marrow cell isolation (Figure 1)
Note: Maintain sterile technique throughout the procedure to prevent contamination. Adjust centrifugation speed and time if using a different centrifuge model. Always follow institutional safety guidelines when handling biological materials.
Bone Marrow Extraction:
a. Place tibias and femurs in a Petri dish on ice.
b. Using scissors, cut both ends of each femur.
c. Transfer the cut femurs to a Petri dish containing complete medium.
Cell Flushing:
a. Hold a cut femur with one hand.
b. Prepare a 10 mL syringe with a 26 G needle, filled with 5 mL of cold sRPMI.
c. Insert the needle into one end of the cut femur.
d. Slowly flush the sRPMI through the femur, collecting the cells in the Petri dish.
e. Repeat for all femurs.
Cell Straining:
a. Place a 70 μm cell strainer on top of a 50 mL tube.
b. Transfer the bone marrow cell suspension onto the strainer.
c. Using a syringe plunger, gently press the cells through the strainer.
d. Rinse the strainer with sRPMI, adding up to a total volume of 10 mL.
e. Discard the strainer, retaining the filtered cell suspension in the tube.
Cell Pelleting:
a. Centrifuge the cell suspension at 1800 rpm for 5 minutes at 4°C.
b. Carefully aspirate and discard the supernatant.
c. Gently resuspend the cell pellet in the remaining liquid.
Cell Counting and Viability Assessment:
a. Add 1 mL of sRPMI to the resuspended cells.
b. Prepare a 1:1 dilution of the cell suspension with Trypan blue.
c. Load the diluted sample onto a hemocytometer.
d. Count viable (unstained) and non-viable (blue-stained) cells.
e. Calculate the total cell count and viability percentage.
Sample Preparation for Flow Cytometry:
a. Determine the number of cells required for flow cytometry staining.
b. Aliquot the appropriate volume of cell suspension for immediate use in flow cytometry staining, preferably in a 96-well U-bottom plate.
c. Store the remaining cells at 4°C as per experimental requirements.
Flow cytometry staining
Bone marrow cells are stained in 96-well plates, which minimizes cell loss during washing and staining steps.
Note: Fluorochrome combinations should be carefully selected based on the instrument and available lasers. Consultation with flow cytometry core personnel is strongly recommended to minimize spectral overlap. All antibodies should be titrated to determine the optimal staining concentration.
Viability staining:
a. Wash the transferred BM cells: add PBS1x to each sample up to 200ul and centrifuge the cell suspension in the 96-well U-bottom plate, 300g for 5 minutes at 4°C.
b. After centrifugation, discard the supernatant by pouring the contents of the plate in a single step, minimizing cell loss.
c. Add the adequate viability staining following the manufacturer recommendations. E.g. LIVE/DEAD™ Fixable Aqua Dead Cell Stain Kit.
d. Incubate 30 minutes at 4°C.
e. Wash the cells adding more PBS1x and centrifugate 300g for 5 minutes at 4°C. After centrifugation, discard the supernatant by pouring the contents of the plate in a single step.
Blocking step (reccomended)
a. After the final wash, resuspend the pellet in 100ul of 1× PBS containing 1% BSA to block nonspecific interactions, and incubate for 15–20 minutes at 4 °C.
Note: During this incubation time, it is recommended to prepare the antibody cocktail (step 3.3) and keep it on ice until use.
b. Wash the cells adding 100ul of PBS 1X, discard the supernatant by pouring in a single step, and proceed with the staining.
Prepare antibody cocktail:
a. Add anti-mouse CD19 APC-Cy7, anti-mouse B220/CD45R FITC, anti-mouse CD43 AF700, anti-mouse CD45 PE-Texas Red, anti-mouse IgM PerCP-Cy7, and anti-mouse IgD PE at appropriate concentrations in FACS buffer. The fluorochrome combination listed below was optimized for use on cytometers equipped with at least three lasers: blue (488 nm), red (640 nm), and violet (405 nm).
Add the complete antibody cocktail prepared in step 3.3 to each sample.
a. For each antibody prepare additional control samples: no stain, single antibody staining, multi-antibody staining, and fluorescence minus one (FMO) staining controls.
b. Incubate in the dark for 30 minutes, RT.
c. Add wash buffer and centrifuge the sample at 800×g for 5 min at RT.
d. Wash twice with wash buffer and resuspend with FACS. Prepare samples for run in the cytometer.
Flow cytometry
Flow cytometry procedure
The no stain sample is used to set laser voltages, the single-color staining samples are used to compensate fluorochrome emission overlap, and the FMO samples are used to provide empirical evidence that the instrument was well compensated. Instrument compensation is essential for proper interpretation of the results.
Run and record all samples, including all controls, after compensation adjustment.
Data analysis using FlowJo software
Gating guideline
This gating strategy enables the resolution of distinct developmental stages of B lymphocytes in the mouse bone marrow, from early progenitors to mature B cells. (Figure 2)
Bone marrow cells were first gated based on forward scatter (FSC-A) and side scatter (SSC-A) properties to select the lymphocyte-enriched population.
Doublets were excluded by FSC-A versus FSC-H gating, and viable cells were identified using the viability dye (Live Aqua, negative gate), first panel in the figure below.
After singlets selection, hematopoietic cells were then defined as CD45⁺.
Within the CD45⁺ fraction, B-lineage cells were identified as CD19⁺B220+ and early progenitor B cells (Pre-Pro B) were defined as CD19-B220 ⁺.
Progression to more differentiated stages was assessed by surface IgM and IgD expression: immature B cells were defined as IgM⁺IgD⁻, whereas mature B cells were identified as IgMlow/⁺IgD⁺. Second panel in the figure.
In the third panel, inside the CD45⁺ fraction, B-lineage cells were further subdivided into Pre, Pro, and Pre-Pro B populations according to CD43 and B220 expression. Additional exclusion of IgD+ and IgM+ cells eliminates contaminating immature and mature B cells.
Protocol references
Bleesing, J. J. H., & Fleisher, T. A. (2002). Human B cells express a CD45 isoform that is similar to murine B220 and is downregulated with acquisition of the memory B-cell marker CD27. Cytometry Part B: Clinical Cytometry, 51(1), 1–8. https://doi.org/10.1002/cyto.b.10007.
Sato, S., Hasegawa, M., Fujimoto, M., & Takehara, K. (2004). Altered blood B lymphocyte homeostasis in systemic sclerosis: expanded naive B cells and diminished but activated memory B cells. Arthritis & Rheumatism, 50(6), 1918–1927. https://doi.org/10.1002/art.20274
Wells, S. M., Stall, A. M., & Kantor, A. B. (1994). CD43 (S7) expression identifies peripheral B cell subsets. The Journal of Immunology, 153(12), 5503–5515. https://doi.org/10.4049/jimmunol.153.12.5503
Acknowledgements
We thank the staff of the animal facilities at UAEU for their support and assistance in animal handling. We also acknowledge ADSCC for technical support and resources contributing to this work.
