Jun 16, 2025
Ferric ferricyanide reduction test or Schmorl’s stain in modification of Lillie (diffuse endocrinocytes)
- Egor Kuzmin1
- 1Sechenov University
- European Society for Pigment Cell Research Community

Protocol Citation: Egor Kuzmin 2025. Ferric ferricyanide reduction test or Schmorl’s stain in modification of Lillie (diffuse endocrinocytes). protocols.io https://dx.doi.org/10.17504/protocols.io.bp2l6yqm1vqe/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 08, 2025
Last Modified: June 16, 2025
Protocol Integer ID: 219772
Keywords: Endocrinocytes, Schmorl, Lillie, Reaction stain, Melanocyte, Argyrophil, Chromaffin, Endocrine, ferric ferricyanide, ferric ferricyanide reduction test, ferricyanide ion, ferric ion, ferrocyanide reduction technique, melanocyte granule, histochemical method, gastrointestinal endocrine cell, enteroendocrine cell content, numerous other biological substance, diffuse endocrinocyte, stain in modification, cornerstone histochemical method, reducing compound, thyroid colloid, lipofuscin deposit
Abstract
Schmorl's ferrocyanide reduction technique stands as a cornerstone histochemical method for detecting diverse reducing compounds. When ferric ions (Fe³⁺) are reduced to the ferrous state (Fe²⁺), they react with ferricyanide ions to form insoluble Berlin blue. This reductive capacity is exhibited by melanocyte granules, enteroendocrine cell contents, thyroid colloid, lipofuscin deposits, and numerous other biological substances. The method is particularly valuable for comprehensive identification of gastrointestinal endocrine cells. Its key advantages include procedural simplicity and rapid execution compared to argentaffin, argyrophil, and chromaffin techniques.
Guidelines
- Ferric chloride (FeCl₃) may be substituted with ferric sulfate (Fe₂(SO₄)₃).
- Solutions must be prepared immediately before use due to rapid precipitation and subsequent loss of activity.
- Elevated temperature dramatically increases reaction kinetics but significantly reduces the stability of the staining complex.
Materials
- Ferric chloride, FeCl₃
- Potassium ferricyanide, K₃[Fe(CN)₆]
- Hydrogen peroxide, H2O2
- Acetic acid, CH3COOH
- Ortho-xylene, C6H4(CH3)2
- Ethanol 100% or isopropanol 100%
- Distilled water
Troubleshooting
Safety warnings
- Formalin is a volatile compound classified as carcinogenic.
- Xylene, isopropyl alcohol, and ethyl alcohol are volatile substances hazardous to human health, especially following long-term exposure. Procedures utilizing these solvents require a ventilated fume hood and the use of medical gloves (e.g., nitrile).
Before start
Autopsy specimens were fixed by immersion in neutral buffered formalin for 48 hours. Processing involved a standard protocol utilizing a graded series of isopropyl alcohols, followed by paraffin embedding. Histological blocks were cut on a microtome to yield 3-μm sections.Sections were placed on silane-coated adhesive glass slides for secure adhesion. All staining reagents were freshly prepared immediately before staining commenced.
Introduction
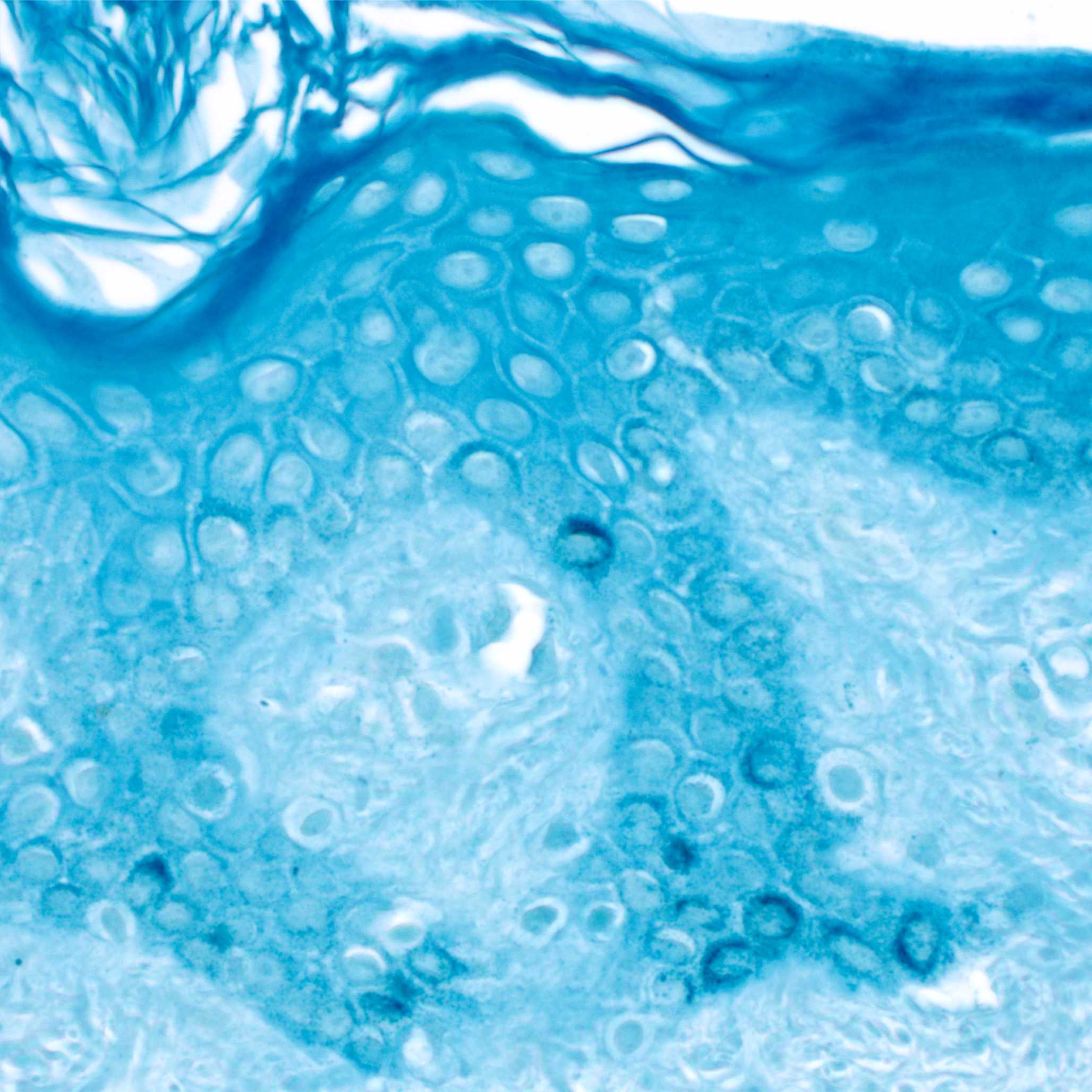
Fig.1. Schmorl's ferricyanide staining on human skin histological sections. In the basal layer of the epidermis, melanin granules within melanocytes are stained. Prior to staining, melanin was decolorized using peroxide bleach.
Preparation of solutions
Working staining solution (Lillie's Modified Solution) 40 mL
Prepare fresh immediately before use
Mix
30 mL of freshly prepared 1% ferric chloride (FeCl₃) solution
4 mL of freshly prepared 1% potassium ferricyanide (K₃[Fe(CN)₆]) solution
6 mL distilled water
Use immediately after preparation. Protect from light. Avoid heating. Unfit for use after several hours.
Аlternative option of the staining solution 100 µL
Mix
75 mL of freshly prepared 1% ferric chloride (FeCl₃) solution
10 mL of freshly prepared 1% potassium ferricyanide (K₃[Fe(CN)₆]) solution
15 mL distilled water
Protocol
1h 15m 30s
STARTING POINT:
Dewaxing, rehydration, rinsing in distilled water. Rinse in pre-warmed distilled water.
- ortho-xylene 5 min
- ortho-xylene 5 min
- absolute ethanol or isopropanol 5 min
- 50% ethanol or isopropanol 5 min
- distilled water 5 min
25m
STAINING:
Treat with 1% hydrogen peroxide for 5 minutes. (Not obligatory)
Used in melanocyte staining to bleach melanin.
After peroxide treatment, rinse sections thoroughly to prevent reaction with the staining solution.
5m
Place sections in the staining solution for 10 minutes at room temperature.
The staining procedure is protocol-specific and may require extended exposure to the staining solution. Staining under minimal light is recommended to prolong reagent shelf life. Some protocol variations permit heating using a water bath, incubator, or microwave oven.
The time for the reaction to take place depends on the substance to be demonstrated, with melanin generally reacting faster than lipofuscin.
10m
Rinse in 1% acetic acid solution.
30s
Nuclear counterstaining. (Not obligatory)
For counterstaining, the original Lillie protocol recommends using an acidified fuchsin solution followed by differentiation in 1% acetic acid.
Alternatively, Nissl's neutral red can be used with differentiation in acidified alcohol.
10m
FINAL STEP:
Rinse, dehydrate, mount in resin medium.
- distilled water 5 min
- 50% ethanol or isopropanol 5 min
- absolute ethanol or isopropanol 5 min
- ortho-xylene 5 min.
- ortho-xylene 5 min.
- moun
25m
Results
Blue staining: melanin, argentaffin cells, chromaffin, some lipofuscins, thyroid colloid and bile
Fig.2. Macrophotograph of bovine small intestine stained by Schmorl's method. Granules of crypt enteroendocrine cells are stained blue.
Protocol references
1. Lillie R, Burtner H. The ferric ferricyanide reduction test in histochemistry. J Histochem Cytochem. 1953 Mar;1(2):87-92. doi:10.1177/1.2.87.
2. Lillie RD, Donaldson PT. The mechanism of the ferric ferricyanide reduction reaction. Histochem J. 1974 Nov;6(6):679-84. doi: 10.1007/BF01011507.
