Oct 23, 2025
Extraction of PCB18 (2,2',5-Trichlorobiphenyl) and Its Hydroxylated Metabolites from Animal Tissues v2
- Xueshu Li1,
- Sean Brobston1,
- Hans-Joachim Lehmler1
- 1University of Iowa

Protocol Citation: Xueshu Li, Sean Brobston, Hans-Joachim Lehmler 2025. Extraction of PCB18 (2,2',5-Trichlorobiphenyl) and Its Hydroxylated Metabolites from Animal Tissues v2. protocols.io https://dx.doi.org/10.17504/protocols.io.14egnrr26l5d/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 22, 2025
Last Modified: October 23, 2025
Protocol Integer ID: 230527
Keywords: PCB18, extraction, tissue, liquid-liquid extraction, polychlorinated biphenyls, hydroxylated polychlorinated biphenyls, PCB, OH-PCB, extraction of pcb18, extracting pcb18, pcb18s in the tissue, hydroxylated metabolites from animal tissue, metabolites from mouse tissue, hydroxylated metabolite, pcb18, levels of pcb18, trichlorobiphenyl, liquid extraction, monohydroxylated metabolite, extraction, gas chromatography, tandem mass spectrometry, mouse tissue, hydroxylated metabolites from animal tissues v2, animal tissues v2
Funders Acknowledgements:
NIEHS
Grant ID: R01ES014901
NIEHS
Grant ID: R01ES031098
NIEHS
Grant ID: R01ES034691
NIEHS
Grant ID: P42ES013661
Abstract
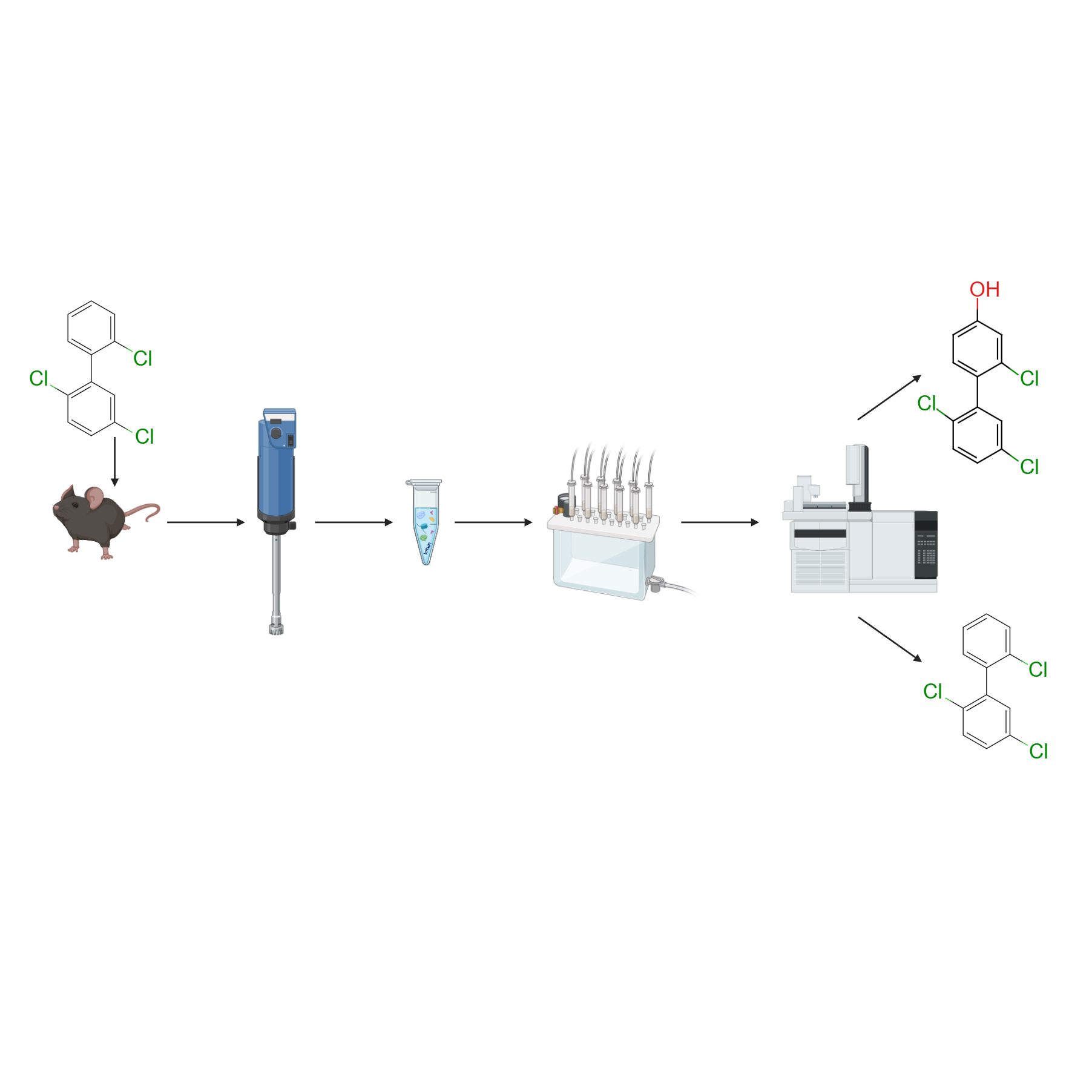
This protocol describes a method for extracting PCB18 (2,2′,5-trichlorobiphenyl) and its monohydroxylated metabolites from mouse tissues via liquid-liquid extraction followed by solid-phase extraction cleanup. The levels of PCB18 and OH-PCB18s in the tissue are subsequently measured by gas chromatography-tandem mass spectrometry.
Materials
Chemicals list
- 2-propanol (Fisher, BPA4164)
- acetone (Fisher, BPA18P4)
- diazomethane (CH2N2) (Hudlicky, 1980; Sigma Aldrich, 2003)
- dichloromethane (DCM), pesticide grade (Fisher, D12-4)
- diethyl ether (Sigma-Aldrich, 673811)
- ethanol (EtOH) (Fisher, A4094)
- hexanes, pesticide grade (Fisher, H300-4)
- methanol (Fisher, A412-4)
- Isooctane, pesticide grade (Fisher, O297-4)
- milli-Q water
- phosphoric acid (H3PO4, Fisher, A242-500)
- silica gel, 150-230 mesh (Fisher, 042727A1)
- sodium chloride (NaCl) (Fisher, BP358-212)
- hydrochloric acid (HCl) (Fisher, S25838)
- potassium chloride (KCl) (Fisher, P217-500)
- sulfuric acid (H2SO4), concentrated (Fisher, A300C-212)
Analytical standards
- d-PCB30 (2,4,6-trichlorobiphenyl-2',3',4',5',6'-d5) (CDN Isotopes, Lot#: X-393)
- PCB18 (2,2',5-trichlorobiphenyl) (Milestone PharmaTech USA Inc., Lot#: MP143P71)
- 4'-OH-PCB18 (2,2',5-trichlorobiphenyl-4'-ol; AccuStandard, Catalog#: HPCB-3004N)
- PCB204 (AccuStandard, Catalog#: C-204N)
- 4-OH-PCB52 (Li and Lehmler, 2022)
Analytical standard solutions
- Surrogate standard (SS)
d-PCB30 (SS_PCB, 100 ng/mL in isooctane)
4-OH-PCB52 (SS_OH-PCB, 100 ng/mL in methanol)
- Internal standard (IS)
PCB204 (100 ng/mL each in isooctane)
- Analytes (included in the OPR)
PCB 18 (100 ng/mL in isooctane)
4'-OH-PCB18 (100 ng/mL in isooctane)
Accessories
- TissueRuptor II (Qiagen, 9002755)
- TissueRuptor Disposable Probes, nonsterile (Qiagen, 990890)
- Serological pipette, 10 mL, (Fisher, 13-678-11E)
- Tube racks
- Disposable Glass Pasteur pipette (short pipette), 5 3/4”, 2 mL (Fisher, 13-678-20A, box of 720)
- Disposable Glass Pasteur pipette (long pipette), 9”, 2 mL (Fisher, 13-678-20C, box of 720)
- Disposable Glass tubes, size 16 x 125 mm (Fisher, 14-959-35A)
- Disposable Glass tube caps (Fisher, 02-883-8D, black screw caps with PTFE liners)
- Repeater pipette (BrandTech, UX-24806-05)
- Syringe Tips NanoRep, 50 mL (Rainin, ENC-50ml)
- SPE Cartridges (Sigma-Aldrich, 52797-U)
- GC vials, 2ml (Fisher, 03-391-6, crimp wide opening autosampler vials)
- Crimp aluminum seal cap, 11 mm (Fisher, 200154, PTFE/silver 100 pack)
- Crimper aluminum seal cap, 11 mm (Fisher, 200154, PTFE/silver 100 pack)
Instruments
- Eppendorf centrifuge 5810, (Eppendorf, 022625004)
- Vortex mixer, (Fisher, 50-728-002)
- Tube rotator, (Fisher, 88-861-122)
- Furnace (Barnstead Thermolyne, 30400)
- AquaTherm Water Bath Shaker, (Stellar, SL-SWB-17)
- N-EVAP analytical evaporator with needles, (Thomas Scientific, 1156Y21)
- SPE Vacuum Manifold, 12 ports, (Sigma-Aldrich, 57-160-U & 57-162-U)
Reagents and solvents
- Hexanes-diethyl ether (9:1, v/v): Measure 90 mL of hexanes and 10 mL of diethyl ether with graduated cylinders, transfer both solvents to an amber bottle, cap, and mix thoroughly.
- 1% KCl (w/w): Weigh 5 g of KCl into a 500 mL volumetric flask, fill to the mark with Milli-Q water, then cap and invert until the salt is completely dissolved.
- 6M HCl: Pipette 50 mL of 12 M HCl into a 100 mL volumetric flask, bring to volume with Milli-Q water, cap, and invert to mix.
- 0.1 M phosphoric acid in 0.9% NaCl solution: Add 0.9 g of NaCl and 1.15 g of 85 % phosphoric acid to a 100 mL volumetric flask, fill to the mark with Milli-Q water, then cap and mix.
- Acidified silica gel: Place silica gel in a clean beaker and combust at 450 °C overnight. Allow it to cool to room temperature, then transfer the combusted silica gel to a screw-capped glass bottle. Prepare acidified silica gel by mixing the combusted silica gel with concentrated sulfuric acid at a 5:1 weight ratio (e.g., 40 g of silica gel with 8 g of sulfuric acid). Shake the mixture vigorously until fully homogenized and no lumps remain. Store the prepared acidified silica gel in a separate screw-capped glass bottle until use.
Troubleshooting
Safety warnings
The following section describes precautions for the safe handling of chemicals used in this protocol. Consult the Safety Data Sheet for additional information regarding all chemicals used in this protocol:
Acetone:
Work under hood. Do not inhale the substance/mixture. Avoid the generation of vapors/aerosols. Keep away from open flames, hot surfaces, and sources of ignition. Take precautionary measures against static discharge. Change contaminated clothing. Preventive skin protection is recommended. Wash hands after
working with substance.
Diazomethane:
Work under a well-ventilated fume hood with sash down. Do not inhale the substance/mixture. Avoid contact with exposed skin. Keep away from open flames, hot surfaces, and sources of ignition. Avoid contact with irregular glass surfaces. Proper PPE is required upon handling, including a lab coat, fresh gloves, and a face shield. Change gloves immediately after handling and discard them into the hazard container.
Dichloromethane (DCM):
Wear personal protective equipment/face protection. Do not get in the eyes, on skin, or clothing. Avoid ingestion and inhalation. Vapors are heavier than air and may spread along floors. Handle product only in an enclosed system or provide appropriate exhaust ventilation. Reacts with aluminum and its alloys.
Diethyl ether:
Avoid contact with skin, eyes, and clothing. Follow good hygiene procedures when handling chemical materials. Follow proper disposal methods. Do not eat, drink, smoke, or use personal products when handling chemical substances. Ground and bond containers when transferring material. Do not get in the eyes, on the skin, or clothing. Empty containers retain product residue (liquid and/or vapor) and can be dangerous. Keep away from heat, sparks, and flame. Do not ingest or inhale. Prevent thebuild-up of vapors to explosive concentration.
Hexanes:
Work under a hood. Do not inhale the substance or mixture. Avoid generating vapors or aerosols.
Hydrochloric Acid:
Do not breathe vapors or aerosols. Avoid substance contact. Ensure adequate ventilation. Evacuate the danger area, observe emergency procedures, and consult an expert.
Methanol:
Use in a chemical fume hood. Wash hands before breaks and immediately after handling the product. Avoid
contact with skin, eyes, and clothing. Take precautions against static discharge.
Methyl tert-butyl ether (MTBE):
Do not breathe vapors or aerosols. Avoid substance contact. Ensure adequate ventilation. Keep away from heat and sources of ignition. Evacuate the danger area, observe emergency procedures, and consult an expert.
Phosphoric acid (H3PO4)
Wash hands after handling. Do not mix with bases. Use in a chemical fume hood. Follow good hygiene. Do not eat, drink, smoke, or use personal products when handling chemical substances. Use only in well ventilated
areas. Prevent contact with eyes, skin, and clothing.
Polychlorinated biphenyls (PCBs):
May cause damage to organs through prolonged or repeated exposure. Very toxic to aquatic life with long-lasting effects. Do not breathe dust/fume/gas/mist/vapors/spray. Avoid release to the environment. This statement does not apply where this is the intended use. Get medical advice/attention if you feel unwell. Collect spillage. Dispose of contents/containers following relevant regulations.
Potassium chloride (KCl):
Minimize dust generation and accumulation. Wash hands after handling. Avoid dispersal of dust in the air (i.e.,
clearing dust surfaces with compressed air). Routine housekeeping should be instituted to ensure that dusts do not accumulate on surfaces. Dry powders can build static electricity charges when subjected to the friction of transfer and mixing operations. Follow good hygiene procedures when handling chemical materials. Do not eat, drink, smoke, or use personal products when handling chemical substances. Avoid generation of dust or fine particulate. Avoid contact with eyes, skin, and clothing.
2-Propanol:
Wear personal protective equipment/face protection. Keep away from open flames, hot surfaces and sources of ignition. Use spark-proof tools and explosion-proof equipment. Use only non-sparking tools. Take precautionary measures against static discharges. Do not get in eyes, on skin, or on clothing. Do not breathe mist/vapors/spray. To avoid ignition of vapors by static electricity discharge, all metal parts of the equipment must be grounded.
Sulfuric acid (H2SO4):
Avoid contact with skin, eyes, and clothing. Follow good hygiene procedures when handling chemical materials. Follow proper disposal methods. Do not eat, drink, smoke, or use personal products when handling chemical substances.
Ethics statement
The responsible Institutional Animal Care and Use Committee (IACUC) approved the animal protocol used to generate tissue samples. All animals were utilized in accordance with all Public Health Service (PHS) policies and the Guide for the Care and Use of Laboratory Animals, National Institutes of Health (NIH) Publication No. 85-23, revised 2010.
Before start
Always wear proper Personal Protective Equipment and work in a fume hood when working with hexanes, hydrochloric acid, MTBE, diazomethane, polychlorinated biphenyl (PCBs) and their derivatives, and strong acids and bases.
Note that this protocol may involve work with experimental animals and requires prior approval by the users' Institutional Animal Care and Use Committee (IACUC) or equivalent ethics committee.
Preparation
Combust the silica gel, glass wool, all glassware (cartridges, short and long pipettes, tubes, beaker/volumetric flask, funnel), scalpel, forceps, and spatula.
Label all sample tubes (Tube-A1, A2, etc.; Tube-B1, B2, etc.; Tube-C1, C2, etc.; Tube-D1, D2, etc., and Reference).
Day 1
Remove the Ongoing Recovery and Precision (ORP) standard (PCB18 and 4'-OH-PCB18) and the Surrogate Standard (SS_PCB, d-PCB30; SS_OH-PCB, 4-OH-PCB52) solutions from the freezer and allow them to warm to room temperature (~ 30 min).
Tissue Homogenization
Place the tissues (e.g., liver ~ 100 mg or brain ~ 100 mg) into tube-A and record the tissue weight.
Note: Wash scalpel, forceps, and spatula between samples with water, acetone, and hexanes.
Add 3 mL of 2-propanol to all samples in tube-A with a repeater pipettor.
Homogenize each sample for 30–60 seconds.
Note: Rinse the TissueRuptor blade between samples with water in a beaker, followed by 2-propanol (~ 5 mL in a test tube).
Add 1 mL of diethyl ether with a repeater pipettor in a fume hood to tube-A, recap, and vortex gently.
Addition of Analytical Standards
Add 100 µL each of d-PCB30 (SS_PCB, 100 ng/mL) and 4-OH-PCB52 (SS_OH-PCB, 100 ng/mL) to all tube-A samples (method blanks, tissue blanks, tissues, OPR, and Reference standard).
- d-PCB30, 100 µL x 100 ng/mL = 10 ng each
- 4-OH-PCB52, 100 µL x 100 ng/mL = 10 ng each
Spike the following analytes only to tube-A OPR samples and the Reference standard.
- 100 µL of PCB18 standard (100 µL x 100 ng/mL = 10 ng)
- 100 µL of 4'-OH-PCB18 standard mixture (100 µL x 100 ng/mL = 10 ng each)
Cap tube-A and the Reference standard.
Put the Reference standard aside until the derivatization step.
Extraction
Note: All centrifugation steps described below are performed for 5 minutes at 3000 rpm (approximately 1811 g-force). Tubes are inverted on the tube rotator for 5 mins at 40 RPM.
Note: For the evaporation or “blow down” step with nitrogen described below, a warm water bath (35 °C) can be used if needed.
Invert tube-A on the tube rotator for 5 minutes at 40 RPM.
Centrifuge Tube-A at 3000 RPM for 5 min.
Add 5 mL of phosphoric acid (H3PO4) in NaCl with repeater pipettor to tube-B.
Transfer the supernatant from tube A to tube B with a long pipette.
Add 1 mL of 2-propanol and 3 mL of hexanes-diethyl ether (9:1, v/v) to tube-A with a repeater pipettor, vortex for 10 secs, and centrifuge.
Transfer the supernatant from tube-A to tube-B.
Invert tube-B for 5 minutes, then centrifuge.
Transfer the organic layer (top layer) from tube-B to a new tube-C with new long pipettes.
Re-extract tube-B with 3 mL of hexanes-diethyl ether (9:1, v/v), vortex, and centrifuge.
Transfer the supernatant from tube-B to combine with the extract in tube-C (~7 mL).
Evaporate solvent in tube-C under a gentle stream of nitrogen to ~100 µL.
Derivatization
Get samples, an ice box, and serological pipettes ready in a fume hood.
Add 5 drops of methanol to tube-C (excluding the Reference standard), vortex for 5 seconds, and place the samples in the fume hood.
Take the diazomethane solution in diethyl ether vial out of the freezer and place it in the ice box in the fume hood.
In the fume hood, using a 10 mL serological pipette, add ~0.5 mL of diazomethane to each sample and 1 mL to the Reference standard.
Cap the tubes and store them in an explosion-proof refrigerator (NOT freezer) at 4-8 °C for at least 3 h or overnight (approximately 16 h).
Day 2
Label GC vials with the sample number.
Remove the Internal Standards (IS) from the freezer and allow to warm to room temperature (~ 30 min).
Derivatization (continued)
Evaporate the excess of ether and diazomethane in a fume hood under a gentle nitrogen flow (no yellow color, ~200 µL).
Evaporate the Reference standard to near dryness (~ 50 μL) and put it aside for the IS spiking step.
Lipid Removal (SPE)
Note: The SPE cartridge can also be prepared using a "dry reagent loading" method. Briefly, glass wool is placed at the bottom of the SPE cartridge, and 0.2 g of silica gel and 2 g of acidified silica gel are added. Then, 3 mL of hexanes-DCM (1:1, v/v) mixture is passed through the cartridge to rinse the filled cartridge.
Note: DCM is toxic and must be handled in a fume hood with adequate ventilation.
Add uncombusted tubes to the bottom of the SPE manifold to collect waste eluent from the SPE cartridge.
Replace the top and place the clean SPE cartridges into each port.
Add glass wool to the SPE cartridge and pack it down with the end of a glass pipette.
Add 0.2 g of silica gel with a combusted small glass funnel to the SPE cartridges.
Add 2 mL of hexanes-DCM (1:1, v/v) with a long pipette to rinse the SPE cartridge.
In a small beaker, mix hexanes-DCM (1:1, v/v) with acidified silica gel (5:1), then add the suspension to the SPE cartridge.
Mix the suspension with a short glass pipette tip to eliminate air bubbles.
Let the silica gel precipitate to the bottom.
Pass ~ 3 mL of hexanes-DCM (1:1, v/v) into the bottom waste tube.
Notes:
- Always ensure the solvent level remains above the acidified silica gel level.
Replace the waste tubes at the bottom with freshly labeled, combusted tube-D. Align tube-C in a test tube rack to match the corresponding tube-D positions in the SPE manifold.
With long glass pipettes, slowly load the extracts from tube-C into the SPE cartridge and pass them through the cartridge for collection.
Rinse tube-C twice (2 x 0.5 mL) with hexanes-DCM (1:1, v/v) and load the wash solvent into the SPE cartridge.
Add additional hexanes-DCM (1:1, v/v; total volume approximately 9 mL) to the SPE cartridge until the eluent in tube-D reaches approximately 10 mL. Then, open the SPE valve completely to flush all the residual solvent into tube-D.
Notes for steps 43-45:
- Maintain a controlled drop-by-drop flow of the eluent by regulating the vacuum pressure of the manifold and SPE valve.
- Always ensure the solvent level remains above the level of the acidified silica gel until the SPE step is complete.
Turn off the manifold vacuum and remove tube-D from the SPE manifold.
Concentrate the samples in tube-D under a gentle stream of nitrogen to ~200 µL.
Add 3 mL of hexanes to tube-D, then continue to concentrate the sample under a gentle stream of nitrogen to ~50 µL to exchange the solvent.
Internal Standard (IS) Spiking and Preparation for Analysis
Add 100 µL of the internal standard (IS; PCB204, 100 ng/mL) to tube-D and the Reference standard using a single-channel pipette.
Vortex tube-D.
Transfer extracts from tube-D and the Reference standard with a long pipette to combusted crimp-style GC vials.
Rinse tube-D with hexanes and combine with the extract in the GC vials to give a final volume of ~0.6 mL.
Prepare the solvent blank by filling a GC vial with hexanes.
Note: Use the same hexanes used for the extractions.
Cap all vials immediately with a crimp cap.
Store samples in a solvent-proof -20°C freezer until instrument analysis.
Note: Do not store samples in a regular -20°C freezer, as solvent vapors will damage the freezer over time.
Disposal and Cleaning
Dispose of the following materials as indicated:
- Scalpel – sharps container.
- Homogenizer probe – dispose as PCB-contaminated hazardous waste.
- SPE cartridges – Remove the cartridges from the SPE manifold and dispose the content as PCB-contaminated hazardous waste. Place the cartridges in the sink to be washed.
- Evaporator – Place needles in a glass beaker and combust in the furnace.
- Tube-A, Tube-B, Tube-C, Tube-D, and other aqueous solutions – dispose as aqueous hazardous waste.
- Organic solvents (waste tubes with hexanes and 2-propanol) – dispose as organic hazardous waste.
- All PCB-exposed disposable glassware, foil, GC vials, and gloves – dispose as PCB-contaminated hazardous waste.
- Serological pipettes – sharps container.
Cleaning:
- The used glass cartridges are washed with soapy water and a brush. After drying, the cartridges are combusted in the furnace.
- The used needles are combusted in the furnace.
Archiving all Samples
Recap all samples after instrument analysis to minimize solvent evaporation. If necessary, refill the solvent to a final volume of ~0.6 mL.
Store all samples, including the solvent blank, in a solvent-proof -20°C freezer until the data are published.
Note: Do not store samples in a regular -20°C freezer, as solvent vapors will damage the freezer over time.
Data Analysis
Use the Excel worksheet included with this protocol to calculate the levels of PCB18 and its hydroxylated metabolites after gas chromatographic analysis.
Protocol references
Hudlicky, M. An improved apparatus for the laboratory preparation of diazomethane. J Org Chem 1980, 45, 5377.
Li, X. S.; Lehmler, H.-J. Dataset for synthesis and authentication of 2,2',5,5'-tetrachlorobiphenyl-4-ol (4-OH-PCB 52). University of Iowa (dataset) 2022. https://doi.org/10.25820/data.006178
Sigma Aldrich, 2003, “AL-180: Diazald, MNNG and diazomethane generators. http://www.sigmaaldrich.com/aldrich/bulletin/AL-180.pdf
General references describing similar extraction protocols
Li X.S., Hefti M.M., Marek R.F., Hornbuckle K.C., Wang K., Lehmler H.J. Assessment of polychlorinated biphenyls and their hydroxylated metabolites in postmortem human brain samples: Age and brain region differences. Environ Sci Technol 2022, 56(13):9515-9526.
Li X.S., Breese, N.M., Lehmler H.J. 2024, Liquid-liquid extraction of 3,3'-dichlorobiphenyl (PCB11) and its hydroxylated metabolites from animal tissues. protocols.io https://dx.doi.org/10.17504/protocols.io.36wgq3w8xlk5/v1
Marek R. F.; Thorne P. S.; DeWall J.; Hornbuckle K. C. Variability in PCB and OH-PCB serum levels in children and their mothers in urban and rural U.S. communities. Environ Sci Technol 2014, 48, 13459–13467.
Milanowski B.; Lulek J.; Lehmler H.-J.; Kania-Korwel I. Assessment of the disposition of chiral polychlorinated biphenyls in female Mdr 1a/b knockout versus wild-type mice using multivariate analyses. Environ Int 2010, 36, 884–892.


