Oct 30, 2025
Epon embedding and ultrathin sectioning of spheroids for transmission electron microscopy
Peer-reviewed method
- Larisa Tratnjek1,
- Aleksandar Janev1,
- Nataša esnik1,
- Uroš erkvenik1,2,
- Mateja Erdani Kreft1
- 1Institute of Cell Biology, Medical Faculty, University of Ljubljana;
- 2Department of Biology, Biotechnical Facutly, University of Ljubljana
- PLOS ONE Lab ProtocolsTech. support email: [email protected]

External link: https://doi.org/10.1371/journal.pone.0342659
Protocol Citation: Larisa Tratnjek, Aleksandar Janev, Nataša esnik, Uroš erkvenik, Mateja Erdani Kreft 2025. Epon embedding and ultrathin sectioning of spheroids for transmission electron microscopy. protocols.io https://dx.doi.org/10.17504/protocols.io.4r3l21opjg1y/v1
Manuscript citation:
Tratnjek L, Janev A, Resnik N, Cerkvenik U, Kreft ME (2026) Integrated light and electron microscopy workflow for morphological, molecular and ultrastructural analysis of spheroids. PLOS One 21(3). doi: 10.1371/journal.pone.0342659
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: August 01, 2025
Last Modified: October 30, 2025
Protocol Integer ID: 223867
Keywords: Spheroids, three-dimensional in vitro model, transmission electron microscopy (TEM), epon embedding, ultrasectioning, ultrastructure, SV-HUC-1, T24, bladder cancer, high-resolution imaging, Spheroids, three-dimensional in vitro model, transmission electron microscopy (TEM), epon embedding, ultrasectioning, ultrastructure, SV-HUC-1, T24, bladder cancer, high-resolution imaging, high resolution tem imaging of spheroid cellular ultrastructure, spheroid cellular ultrastructure, ultrathin sectioning of spheroid, spheroids from other cell type, preparing spheroid, using spheroid, vivo tumour cell behaviour, human urothelial cell line, replicating tumour microenvironment characteristic, dimensional cell culture, tumour microenvironment characteristics such as hypoxic core, spheroid, transmission electron microscopy, dimensional spheroid model, electron microscopy, tumour microenvironment, cancer research, relevant system for cancer research, tissue origin, high resolution tem imaging, ultrathin sectioning, cell, other cell ty
Funders Acknowledgements:
Slovenian Research and Innovation Agency ARIS
Grant ID: P3-0108
Slovenian Research and Innovation Agency ARIS
Grant ID: J7-2594
Slovenian Research and Innovation Agency ARIS
Grant ID: MRIC UL IP-0510 Infrastructure Program
Abstract
Three-dimensional spheroid models offer a more physiologically relevant system for cancer research compared to traditional two-dimensional cell cultures by better replicating tumour microenvironment characteristics such as hypoxic cores and acidic pH environments, which activate survival pathways and closely mirror in vivo tumour cell behaviour. Due to their small size and delicate nature, spheroids pose challenges for sample handling and ultrastructural analysis.
This protocol provides a detailed, stepwise procedure for preparing spheroids by epon embedding and ultrathin sectioning, optimized for transmission electron microscopy (TEM). The workflow is demonstrated using spheroids derived from normal (SV-HUC-1) and cancerous (T24) human urothelial cell lines but can be readily adapted to spheroids from other cell types and tissue origins. This method enables consistent, high resolution TEM imaging of spheroid cellular ultrastructure, facilitating in-depth morphological analysis.
Image Attribution
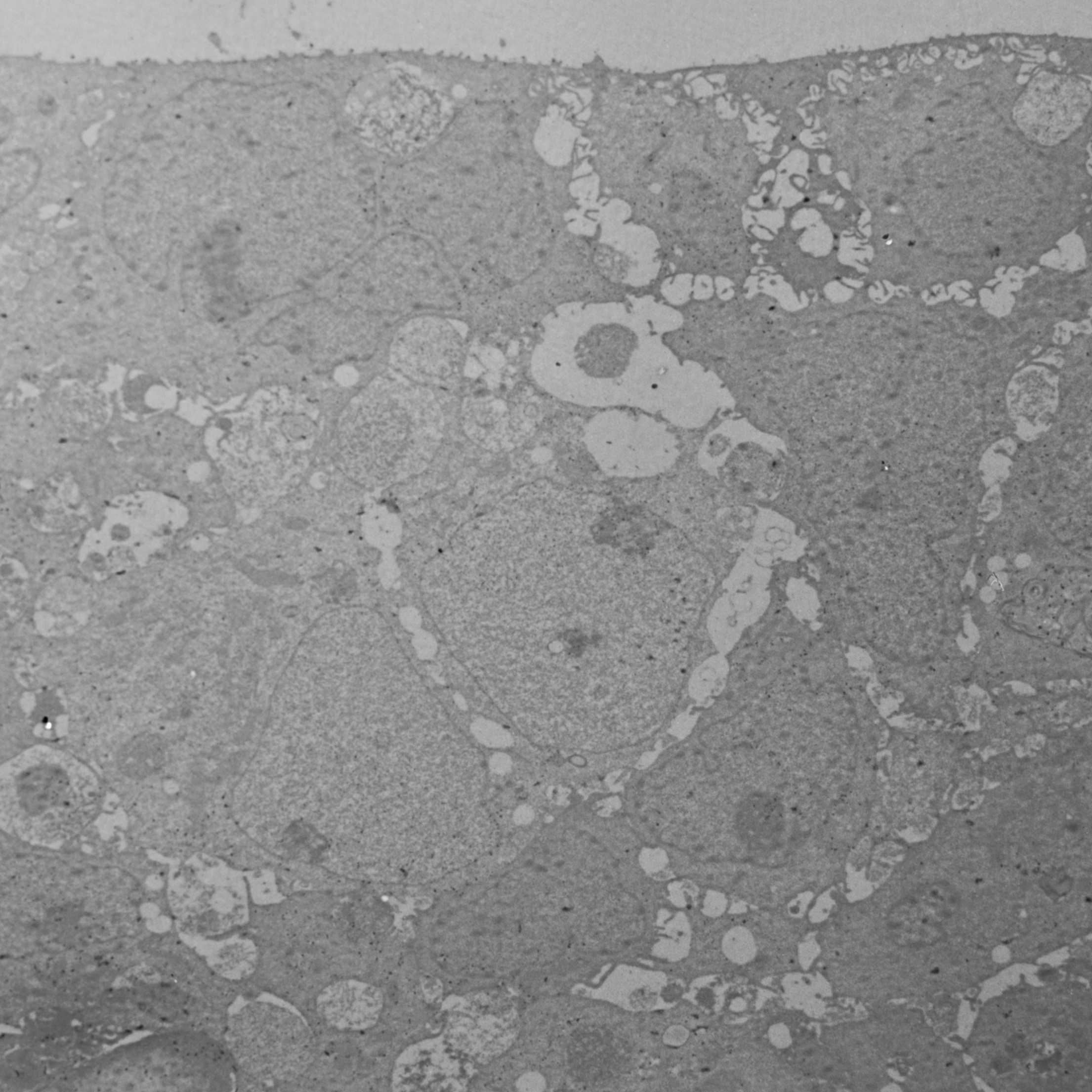
Fig 1. Resin (Epon) embedding of spheroids for TEM. A: Silicon flat embedding moulds with varying cavity sizes for Epon resin. B: Epon resin block with SV-HUC-1 spheroid before and after the trimming (right).
Materials
Materials
- 1 mL and 200 µL micropipette tips (Brand, Germany, Cat. No.: 732012 and 732028)
- 10 mL glass vials (SGD Pharma, Germany)
- Bovine Serum Albumin (BSA) (Sigma-Aldrich, USA, Cat. No.: A2153)
- Cacodylic acid sodium salt trihydrate (Serva, Germany, Cat. No.:1554002)
- Microcentrifuge tubes 0.5 mL and 1.5 mL (LLG Labware, United Kingdom, Cat. No.: 9409026 and 6.490 852)
- Distilled water (Institute of cell biology, ULMF, Slovenia)
- Epon:
- Glycid ether 100,
- Methylnadic anhydride,
- 2-Dodecenylsuccinic acid anhydride,
- 2,4,6-Tris(dimethylaminomethyl)phenol (Serva, Germany, Cat. No.: 21045, 29452, 20755, 36975)
- Ethanol (100%) (Carlo Erba Reagents, France, Cat. No.: 4146072)
- Glutaraldehyde (Serva, Germany, Cat. No.: 23114.01)
- Lead citrate:
- Lead (III) nitrate (Merck, Germany, Cat. No.: 1.07398)
- Tri-Sodium citrate dihydrate (Merck, Germany, Cat. No.: 1.06447)
- Osmium tetroxide (Roth, Germany, Cat. No.:8371.3)
- Paraformaldehyde (Sigma-Aldrich, Germany, Cat. No.: 158127)
- Pasteur pipette (3 mL) (Brand, Germany, Cat. No.:747765)
- Phosphate buffer saline (PBS):
- Potassium chloride (Merck, Germany, Cat. No.:104936),
- Disodium hydrogen phosphate (Merck, Germany, Cat. No.:106580),
- Potassium dihydrogen phosphate (Merck, Germany, Cat. No.:104873),
- Sodium chloride (Chem-Lab, Belgium, Cat. No.:CL00.1429)
- Potassium hexacyanoferrate (II) trihydrate (Merck, Germany, Cat. No.:14459-95-1)
- Silicon moulds 11-039 and 11-035 (Balzers Union, Liechtenstein)
- Sterile 96-well ultra-low attachment U-shaped bottom microplates (Corning, USA, Cat. No.: 7007)
- Sucrose (Sigma-Aldrich, Germany, Cat. No.: S9378)
- Transmission electron microscopy grids (Micro to Nano BV, Netherlands)
- Uranyl acetate (Merck, Germany, Cat. No.:8473)
Equipment
- Bench-mounted fume cupboard System Delta 30 (Wesemann, Germany)
- Centrifuge MyFuge Mini (Benchmark Scientific, USA)
- Laminar air-flow cabinet M182 (II) (Iskra Pio, Slovenia)
- Scale AE 163 (Mettler Toledo, Switzerland)
- Transmission electron microscope CM100, equipped with AMT camera (Philips, The Netherlands)
- Ultramicrotome EM UC6 (Leica, Germany)
Troubleshooting
Protocol for the formation of normal and cancer urothelial spheroids
1w
SV-HUC-1 and T24 spheroids were grown in 96-well ultra-low attachment U-shaped bottom microplates (Corning, New York, NY, USA) and incubated in a humidified incubator at 5% CO2 and 37°C. Seeding
densities of 100,000 cells per well (in 200 µL of culture medium) were used to generate spheroids. Spheroids were grown for 7 days prior to their preparation for transmission electron microscopy analysis.
Note
During seeding, ensure that pipette tips do not touch the bottom or sides of the wells to avoid damaging the surface coating of the ultra-low attachment U-shaped bottom microplates.
Note
Spheroid loss during washing, solution exchanges, and procedures such as dehydration is expected and correlates with operator experience level and handling technique precision. Always process 20–30% excess spheroids beyond experimental requirements to compensate for potential losses.
1w
Spheroid transfer and collection
30m
Room temperature
2.1 Use a 1 mL micropipette fitted with a tip or a 3 mL plastic Pasteur pipette to transfer the spheroids from the 96-well ultra-low attachment U-bottom cell culture microplate to a 1.5 mL microcentrifuge tube filled with cold 4% formaldehyde (w/v) and 2% glutaraldehyde (v/v) in 0.1 M cacodylate buffer (pH 7.2-7.4).
Note
To prevent spheroids from adhering to the pipette wall and to ensure maximal transfer, pre-coat the micropipette tips or Pasteur pipette with 1% BSA in PBS by dipping their full length of the tip in 1% BSA in PBS.
2.2 Quickly aspirate the culture medium and spheroid with the pipette. You should be able to see the spheroid inside the tip. If it is not there, return the medium to the well and aspirate again until you can visually confirm the presence of the spheroid.
2.3 Allow the spheroid to settle at the bottom of the tip, then pipette it into the microcentrifuge tube containing fixative.
Note
You can transfer several spheroids (3-5) into a single microcentrifuge tube or work with individual spheroids.
30m
Fixation
1h 37m
3.1 Fix the spheroids with cold 4% formaldehyde (w/v) and 2% glutaraldehyde (v/v) in 0.1 M cacodylate buffer (pH 7.2-7.4) for 30 minutes at Room temperature ensuring that the spheroids are completely submerged in the fixative.
3.2 Rinse the samples with 0.33 M sucrose in 0.1 M cacodylate buffer for three consecutive 10-minute intervals at Room temperature .
Note
To change the solution in which the spheroid is submerged in a microcentrifuge tube, gently aspirate as much of the solution as possible with a 200 µL/1 mL micropipette with a fitted tip without disturbing the spheroid and add the appropriate volume of the new solution. However, to prevent accidental spheroid loss, maintain 10-20µL of residual liquid at the container bottom during solution exchanges.
Note
When performing solution exchanges, first allow spheroids to settle at the container bottom. For accelerated settling, tubes may be gently spun at 2000×g (6000 rpm) for 5 seconds to ensure complete spheroid sedimentation. Visually confirm the presence of spheroids after each step.
3.3 Transfer the samples to glass vials.
3.4 Post-fix the samples in mixture of 1% (w/v) osmium tetroxide and 0.8% potassium-ferrocyanide (v/v) in 0.2M cacodylate buffer for 30 minutes in the dark, at Room temperature .
3.5 Rinse samples with 0.1 M cacodylate buffer for 5 minutes at Room temperature .
3.6 Rinse samples in distilled water for 2 minutes at Room temperature .
1h 37m
Dehydration
25m
Room temperature
4.1 Dehydrate the samples by incubating in 50% ethanol for 5 minutes.
4.2 Dehydrate the samples by incubating in 70% ethanol for 5 minutes.
4.3 Dehydrate the samples by incubating in 90% ethanol for 5 minutes.
4.4 Dehydrate the samples by incubating in 100% ethanol twice for 5 minutes.
25m
Epon resin embedding
1d 1h
5.1 Transfer the spheroids with spatula or Pasteur pipette and immerse them into a mixture of Epon resin and ethanol (ratio 1:1) in silicon resin embedding moulds with larger cavity sizes (Fig 1) for 1 hour atRoom temperature .
5.2 Transfer and immerse the samples in Epon resin in silicon embedding moulds with smaller cavity sizes (Fig 1), optimal for subsequent sectioning (transfer using a spatula).
5.3 Perform the polymerization of Epon over the next 5 days with gradual temperature increase every 24 hours (35 °C, 45 °C, 60 °C, 70 °C, 80 °C).
Fig 1. Resin (Epon) embedding of spheroids for TEM. A: Silicon flat embedding moulds with
varying cavity sizes for Epon resin. B: Epon resin block with SV-HUC-1 spheroid before and after the trimming (right).
1d 1h
Preparation of semithin and ultrathin sections
6.1 Prepare 1 µm semithin sections to localise spheroids (cells) with the light microscope.
6.2 Cut the 60 nm ultrathin sections and mount them onto grids for transmission electron microscopy.
6.3 Contrast-enhance the ultrathin sections with a saturated solution of uranyl acetate for 20 minutes and a 10% solution of lead citrate for 3 minutes.
Acknowledgements
We thank Sanja Čebraja, Nada Pavlica Dubarič, Marko Radanović and Sabina Železnik for their technical support and expertise in preparing spheroids and samples for electron microscopy. We also thank Urška Dragin Jerman, PhD, for her expertise and assistance with confocal microscopy. The research was done using the Research infrastructure ELIXIR-SI (https://elixir-slovenia.org), funded by the European Regional Development Fund, the Ministry of Science, Education and Sports and by the Slovenian Research and Innovation Agency.
