Oct 07, 2025
Enzyme-linked immunosorbent assay using the SARS-CoV-2 membrane glycoprotein analog M1s (sequence: ac-CTITVEELKKLLEQC-am) for detection of cognate COVID-19 antibodies as performed on high binding plates
- Brian Andrich Pollo1,
- Danica S. Ching2,1,
- Maria Isabel C. Idolor1,
- Ruby Anne King3,
- Fresthel Monica M. Climacosa4,1,
- Salvador Eugenio C. Caoili1
- 1Biomedical Innovations Research for Translational Health Science (BIRTHS) Laboratory, Department of Biochemistry and Molecular Biology, University of the Philippines Manila;
- 2Institute of Clinical Epidemiology, National Institutes for Health, Manila;
- 3Department of Science and Technology - Philippine Council for Health Research and Development (DOST-PCHRD);
- 4Department of Medical Microbiology, College of Public Health, University of the Philippines Manila
- Brian Andrich Pollo: corresponding author;
- Coronavirus Method Development Community

External link: https://doi.org/10.6084/m9.figshare.30276484.v1
Protocol Citation: Brian Andrich Pollo, Danica S. Ching, Maria Isabel C. Idolor, Ruby Anne King, Fresthel Monica M. Climacosa, Salvador Eugenio C. Caoili 2025. Enzyme-linked immunosorbent assay using the SARS-CoV-2 membrane glycoprotein analog M1s (sequence: ac-CTITVEELKKLLEQC-am) for detection of cognate COVID-19 antibodies as performed on high binding plates. protocols.io https://dx.doi.org/10.17504/protocols.io.36wgqpezyvk5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 06, 2025
Last Modified: October 07, 2025
Protocol Integer ID: 229142
Keywords: ELISA, SARS-CoV-2, COVID-19 antibodies, peptide antigen, membrane glycoprotein, serology, immunoassay, Protein A–peroxidase, TMB substrate, seroprevalence, immune monitoring, assay development, synthetic peptide, antibody detection, detecting antibody, using synthetic peptide antigen, linked immunosorbent assay, synthetic peptide antigen, peptide antigen in carbonate, immunosorbent assay, antibodies against the sar, antigen, antibody level, distinct absorbance profiles proportional to antibody level, immune response monitoring, synthetic peptide sequence, incubation with diluted serum, elisa, validation for sar, diluted serum
Funders Acknowledgements:
Dissertation grant
IDC211 grant
Abacov project
NIH faculty grant
CIP fellowship
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
This protocol describes a peptide-based indirect enzyme-linked immunosorbent assay (ELISA) for detecting antibodies against the SARS-CoV-2 membrane glycoprotein analog M1s (synthetic peptide sequence: ac-CTITVEELKKLLEQC-am). High-binding polystyrene plates are coated overnight with the peptide antigen in carbonate-bicarbonate buffer, followed by blocking, incubation with diluted serum, detection using Protein A–peroxidase, and colorimetric readout with TMB substrate at 450 nm. The method offers a simple, reproducible, and cost-effective workflow for COVID-19 serological testing using synthetic peptide antigens. Expected results include distinct absorbance profiles proportional to antibody levels, supporting applications in seroprevalence analysis, immune response monitoring, and assay validation for SARS-CoV-2 antibody research.
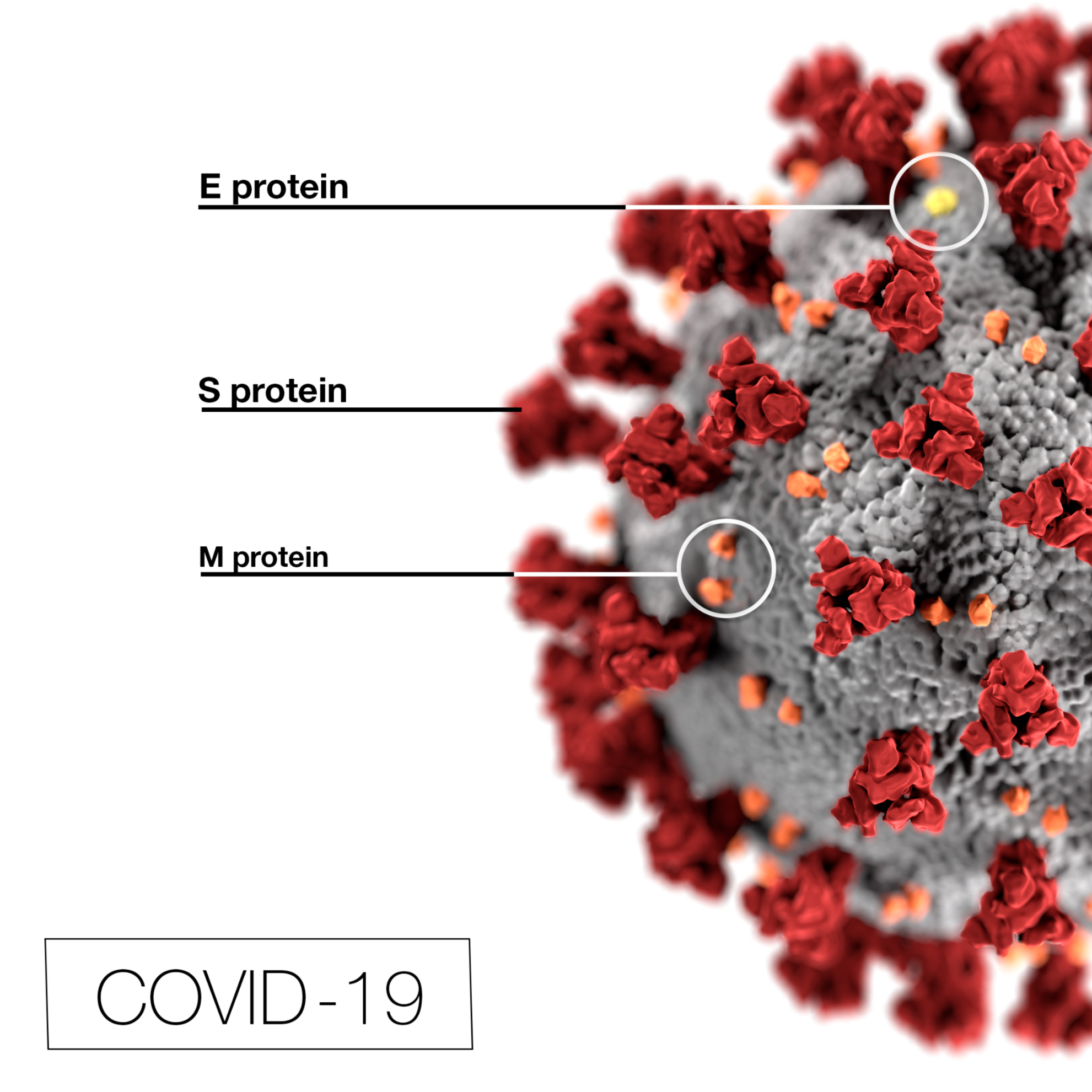
Image Attribution
CDC/ Alissa Eckert, MS; Dan Higgins, MAM, Public domain, via Wikimedia Commons
Troubleshooting
Coat
overnight incubation at 4°C
100μL/well, Carbonate-bicarbonate buffer in water, pH 9.6
Wash
3x at 5 min interval
0.05% Tween 20 in 1X PBS (PBST)
freeze
Block
*leave plates at RT for 15min before blocking*
incubation at 37°C for 30 mins
200μL/well, 5% skimmed milk in PBST
Wash
3x at 5 min interval
0.05% Tween 20 in 1X PBS
Primary antibody
incubation at RT for 1hr
100μL/well, 1:100 sera in dilution buffer (0.05% skimmed milk in PBST)
Wash
3x at 5 min interval
0.05% Tween 20 in 1X PBS
Secondary antibody
incubation at RT for 1hr
50μL/well, 1:2000 Protein A-peroxidase (0.5ug/mL) in dilution buffer
Wash
3x at 5 min interval
0.05% Tween 20 in 1X PBS
Chromogenic substrate solution
incubation at RT for 30 min
50μL/well, 0.1mg/mL TMB in Phosphate Citrate Buffer in water, pH 6.0
Stop solution
50μL/well, 1M H2SO4
read at 450nm
