Aug 19, 2025
Comparative of aerobic vs. isometric exercise on menstrual pain
- Mamta Mandal1,
- K M Amran Hossain2
- 1Mamta Mandal;
- 2Department of Physiotherapy & Rehabilitation, Jashore University of Science & Technology
- Jashore University of Science and Technology

Protocol Citation: Mamta Mandal, K M Amran Hossain 2025. Comparative of aerobic vs. isometric exercise on menstrual pain. protocols.io https://dx.doi.org/10.17504/protocols.io.261ge8q9wg47/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 24, 2025
Last Modified: August 19, 2025
Protocol Integer ID: 220930
Keywords: Aerobic exercise, isometric exercise, VAS, Young females, Progesterone levels , managing menstrual pain, menstrual pain this protocol, reducing primary dysmenorrhea, menstrual pain, primary dysmenorrhea in young female, using plasma progesterone level, menstrual symptoms questionnaire, modified menstrual symptoms questionnaire, plasma progesterone level, exercise
Funders Acknowledgements:
Department of Physiotherapy and Rehabilitation
Grant ID: Jashore University of Science and Technology
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
This protocol outlines a randomized clinical trial comparing the effectiveness of aerobic and isometric exercises in reducing primary dysmenorrhea in young females aged 18–25. Participants will be divided into two groups—two intervention (aerobic and isometric exercise) —with exercises performed thrice weekly for 8 weeks. Expected outcomes will be assessed using plasma progesterone levels, Visual Analog Scale, modified Menstrual Symptoms Questionnaire, and Quality of Life measures. The study aims to identify a more effective non-pharmacological exercise intervention for managing menstrual pain and reducing reliance on medication.
Guidelines
1. Confirm participants meet inclusion criteria and have given informed consent.
2. Check all equipment before use; ensure it’s functional and safe.
3. Follow the intervention and assessment steps exactly as planned.
4. Maintain accurate records and participant confidentiality.
5. Any protocol changes must be approved before implementation.
Materials
Visual Analog Scale (VAS):
- Measures pain intensity
- Scale from 0 (no pain) to 10 (worst pain)
Menstrual Symptoms Questionnaire (MSQ):
- Assesses symptoms like nausea, fatigue, mood swings, and back pain
Quality of Life (QOL) Assessment Tool:
- Evaluates physical and emotional wellbeing related to dysmenorrhea
Participant Information Sheet & Consent Form:
- Ensures ethical approval and voluntary participation
Troubleshooting
Safety warnings
1. Stop the session immediately if participants experience severe pain, dizziness, or unusual symptoms.
2. Keep first-aid and emergency support accessible at all times.
3. Follow hygiene and infection control procedures strictly.
4. Participation is voluntary; participants can withdraw anytime without penalty.
Ethics statement
This study involves human participants and does not include experiments with animals. Prior ethical approval was obtained from the Institutional Review Board (IRB) of the Department of Physiotherapy & Rehabilitation at Jashore University of Science and Technology, Bangladesh, under permit number- PTR-JUST/IRB/2025/04/11, dated May 24, 2025. The IRB reviewed the ethical statement checklist, participant information sheet, consent form, hematology and toxicology study, and questionnaire, ensuring compliance with the Declaration of Helsinki, ICH-GCP guidelines, and local regulatory requirements. The study was approved as a randomized clinical trial, with clauses ensuring no involvement of the study team in IRB deliberations, mandatory reporting of serious adverse events, protocol changes, and final study results.
Comparative of aerobic vs. isometric exercise on menstrual pain
Step 1 — Screening, consent, and baseline (Day 0)
- Confirm eligibility and obtain written informed consent.
- Record demographics and menstrual history (cycle length, pain history, medication use).
- Collect baseline outcomes:
- Pain: Visual Analog Scale (VAS)
- Symptoms: Modified Menstrual Symptoms Questionnaire (MSQ)
- Quality of life (QOL) tool
- Blood sample for plasma progesterone (follow lab SOP; collect at your predefined cycle phase)
- Assign unique participant code and create source documents; set up secure data sheet.
- Schedule first intervention session and provide safety instructions (stop if severe pain, dizziness, or unusual symptoms; withdrawal is voluntary).
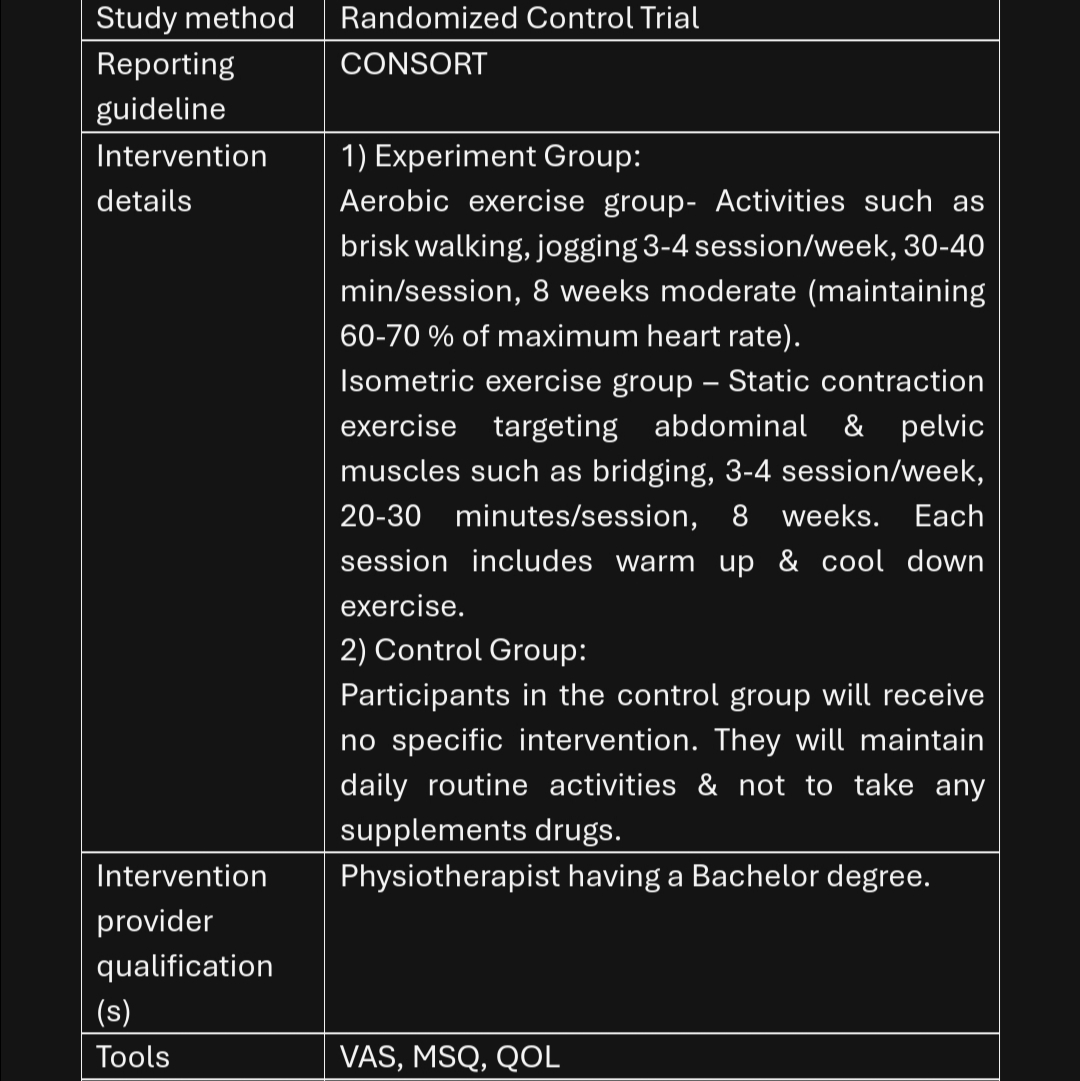
Step 2 — Randomization and 8-week exercise intervention (Weeks 1–8; 3×/week)
- Randomize participant to one arm:
- Aerobic exercise (e.g., treadmill/brisk walking/cycling)
- Isometric exercise (targeted holds for major muscle groups)
- For each supervised session (≈35–45 min):
- Warm-up 5 min of light dynamic movement.
- If Aerobic arm: Continuous aerobic activity at a moderate, steady intensity (define your target here: speed/HR/RPE) for 20–30 min.
- If Isometric arm: Series of isometric holds (define muscle groups, hold duration, sets, rest; keep total working time ~20–30 min).
- Cool-down and breathing/relaxation 5 min.
- Record session adherence, perceived exertion/intensity, and any adverse events in the session log.
- Enforce safety: stop session if severe pain, dizziness, or abnormal symptoms; keep first-aid accessible; follow hygiene and equipment checks each visit.
Step 3 — Outcome assessments and data management (Midpoint & Week 8)
- Mid-program checkpoint (around Week 4): Optional interim VAS/MSQ to monitor trends and safety.
- Post-intervention (Week 8 / predefined cycle window):
- Repeat VAS, MSQ, and QOL.
- Collect post-program plasma progesterone (same lab SOP and cycle phase definition used at baseline).
- Verify completeness and accuracy of all case report forms; de-identify data with participant codes only.
- Enter data into the electronic sheet; audit for errors; lock the database.
- Document any protocol deviations and serious adverse events per IRB requirements; prepare summary for reporting.
Protocol references
- Burnett, M., & Lemyre, M. (2017). No. 345 - Primary dysmenorrhea consensus guideline. Journal of Obstetrics and Gynaecology Canada, 39(7), 585–595. https://doi.org/10.1016/j.jogc.2016.12.023
- Akiyama, S., Tanaka, E., Cristeau, O., Onishi, Y., & Osuga, Y. (2017). Evaluation of the treatment patterns and economic burden of dysmenorrhea in Japanese women, using a claims database. ClinicoEconomics and Outcomes Research, 9, 295–306. https://doi.org/10.2147/CEOR.S127760
- Blakey, H., Chisholm, C., Dear, F., Harris, B., Hartwell, R., Daley, A. J., & Jolly, K. (2010). Is exercise associated with primary dysmenorrhoea in young women? International Journal of Obstetrics and Gynaecology, 117(2), 222–224. https://doi.org/10.1111/j.1471-0528.2009.02220.x
- Kaur, S., Kaur, P., Shanmugam, S., & Kaur Kang, M. (2014). To compare the effect of stretching and core strengthening exercises on primary dysmenorrhea in young females. IOSR Journal of Dental and Medical Sciences, 13(6), 22–32. https://doi.org/10.9790/0853-13652232
- Grandi, G., Ferrari, S., Xholli, A., Cannoletta, M., Palma, F., Romani, C., Volpe, A., & Cagnacci, A. (2012). Prevalence of menstrual pain in young women: What is dysmenorrhea? Journal of Pain Research, 5, 169–174. https://doi.org/10.2147/JPR.S30602
- Golomb, L. M., Solidum, A. A., & Warren, M. P. (1998). Primary dysmenorrhea and physical activity. Medicine & Science in Sports & Exercise, 30(6), 906–909. https://doi.org/10.1097/00005768-199806000-00020
- Ortiz, M. I., Cortés-Márquez, S. K., Romero-Quezada, L. C., Murguía-Cánovas, G., & Jaramillo-Díaz, A. P. (2015). Effect of a physiotherapy program in women with primary dysmenorrhea. European Journal of Obstetrics, Gynecology, and Reproductive Biology, 194, 24–29. https://doi.org/10.1016/j.ejogrb.2015.08.008
- Vaziri, F., Hoseini, A., Kamali, F., Abdali, K., Hadianfard, M., & Sayadi, M. (2015). Comparing the effects of aerobic and stretching exercises on the intensity of primary dysmenorrhea in the students of universities of Bushehr. Journal of Family and Reproductive Health, 9(1), 23–28.
- Munro, C. J., Stabenfeldt, G. H., Cragun, J. R., Addiego, L. A., Overstreet, J. W., & Lasley, B. L. (1991). Relationship of serum estradiol and progesterone concentrations to the excretion profiles of their major urinary metabolites as measured by enzyme immunoassay and radioimmunoassay. Clinical Chemistry, 37(6), 838–844. https://doi.org/10.1093/clinchem/37.6.838
- Iacovides, S., Avidon, I., & Baker, F. C. (2015). Women with dysmenorrhoea are hypersensitive to experimentally induced forearm ischaemia during painful menstruation and during the pain-free follicular phase. European Journal of Pain, 19(6), 797–804. https://doi.org/10.1002/ejp.604
Acknowledgements
We express gratitude to the Department of Physiotherapy & Rehabilitation (PTR) at Jashore University of Science and Technology for partial funding and provision of facilities for this study. We also acknowledge the anticipated contributions of Certified McKenzie Therapists, Orthopedic Spine Surgeons, and the research team for their expertise in executing this future protocol.

