Aug 09, 2025
CAMbank: Body Microbiome Swabbing (v1)
- Eliah Overbey1,2,3,
- Jaume Puig3,
- Jeremy Wain Hirschberg2,
- JangKeun Kim2,3,
- Christopher Mason2,3
- 1University of Austin (UATX);
- 2Weill Cornell Medicine;
- 3BioAstra

Protocol Citation: Eliah Overbey, Jaume Puig, Jeremy Wain Hirschberg, JangKeun Kim, Christopher Mason 2025. CAMbank: Body Microbiome Swabbing (v1). protocols.io https://dx.doi.org/10.17504/protocols.io.j8nlkywnxg5r/v1
Manuscript citation:
Overbey, E.G., Ryon, K., Kim, J. et al. Collection of biospecimens from the inspiration4 mission establishes the standards for the space omics and medical atlas (SOMA). Nat Commun 15, 4964 (2024). https://doi.org/10.1038/s41467-024-48806-z
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: August 08, 2025
Last Modified: August 22, 2025
Protocol Integer ID: 224373
Keywords: microbial sampling, omics, astronaut, spaceflight, body microbiome swabbing, rna swab, standardized collection of body swab, swab collection, comparable microbiome datasets across mission, body swab, swab site, swab, swab tip, microbial communities from multiple anatomical site, microbial diversity analysis, cornell aerospace medicine biobank, microbial community, microbial diversity analyses in both ground, sides of the swab tip, mucosal microbiota, comparable microbiome dataset, rna shield, microbial composition, sterile dna, astronaut health risk assessment, containing dna, moistened with nuclease
Abstract
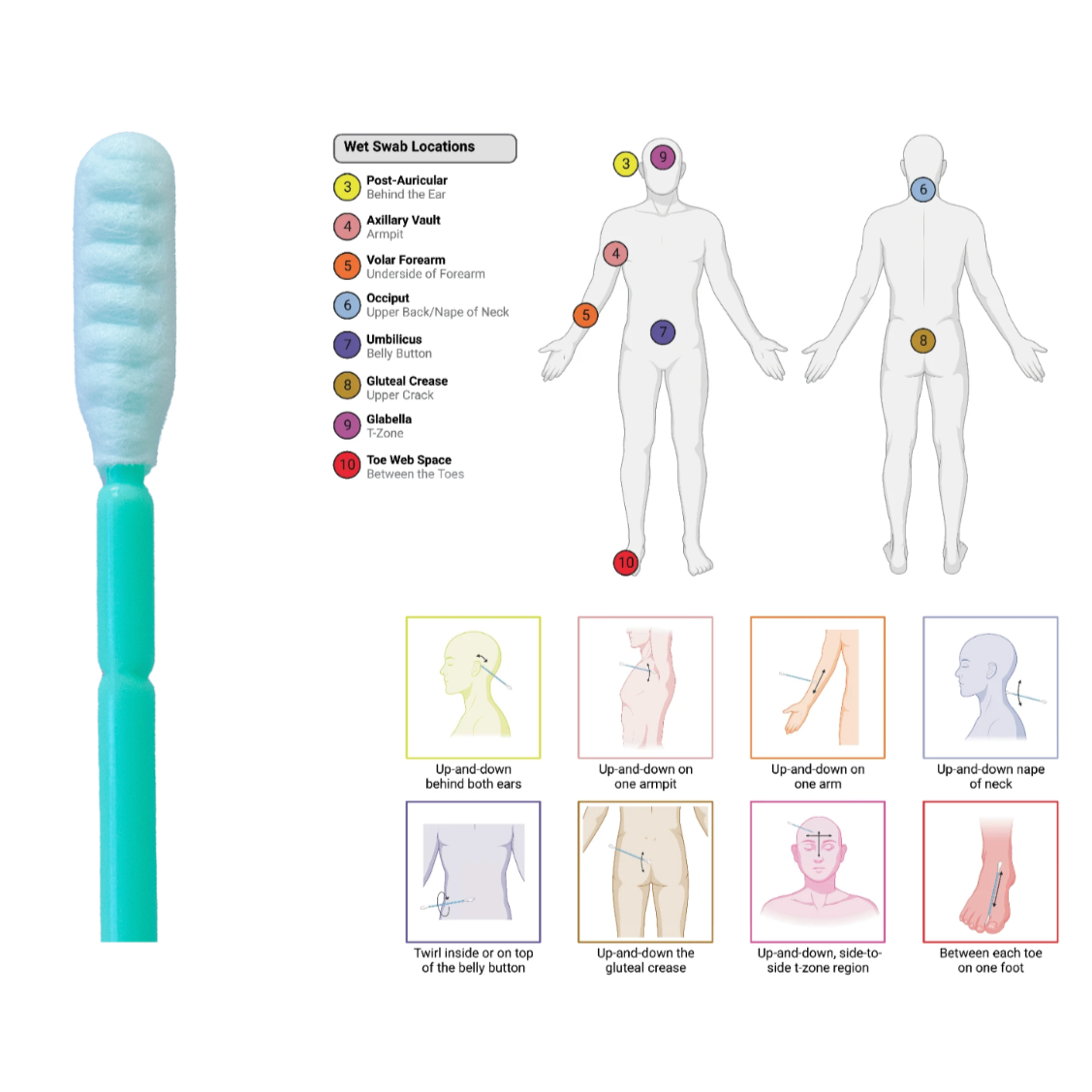
This protocol describes the standardized collection of body swabs for microbiome and microbial diversity analyses in both ground-based and spaceflight settings. The method is designed to capture microbial communities from multiple anatomical sites relevant to astronaut health monitoring. During the SpaceX Inspiration4 mission, swabs were collected at all study timepoints from 10 body locations: two dry-swab sites (oral cavity, nasal cavity) and eight wet-swab sites (post-auricular area, axillary vault, volar forearm, occiput, umbilicus, gluteal crease, glabella, and toe web space). For wet-swab collections, sterile DNA/RNA swabs were pre-moistened with nuclease-free water (ground) or hydrogen-infused water (in-flight). Each site was swabbed for 30 seconds using firm pressure and both sides of the swab tip. Swabs were placed in 2D matrix tubes containing DNA/RNA Shield preservative and stored at 4°C during collection periods. Post-mission, all samples were archived at −80°C in the Cornell Aerospace Medicine Biobank. These swabs are suitable for downstream metagenomic, metatranscriptomic, and targeted amplicon sequencing to assess microbial composition, alpha/beta diversity, and potential shifts in skin and mucosal microbiota associated with spaceflight. The protocol supports the Space Omics and Medical Atlas (SOMA) initiative’s objective of generating standardized, comparable microbiome datasets across missions to inform astronaut health risk assessments and countermeasure development.
Materials
Sterile swabs (Isohelix DNA/RNA Swabs)
Pre-labeled 2D matrix tubes with Zymo DNA/RNA Shield (400 µL per tube/sample)
Sterile nitrile gloves
70% Ethanol in a spray bottle
VWR KimWipes
Cryogenic Freezer
Troubleshooting
Preparation
Check that all materials are available and the swabs are still sterile-sealed.
Put on nitrile gloves to ensure sterile handling of the samples.
Sample Collection
Swab each region of the body.
1. Wet each swab with nuclease-free water (EXCEPT oral and nasal swabs, which do not need to be wetted).
2. Swab the body region for 30 seconds using both sides of the swab.
3. Open the 2D matrix tube and deposit the swab in the Zymo DNA/RNA Shield preservative.
Ensure all tubes are tightly sealed.
Storage
Tube exteriors are sterilized with 70% EtOH and a KimWipe.
Store samples at +4°C (if processing imminently) or at -80°C (for long-term storage).
Acknowledgements
This work was supported by the WorldQuant Foundation, the WayPaver Foundation, and the following NASA grants: NNX14AH50G, NNX17AB26G, 80NSSC22K0254, NNH18ZTT001N-FG2, NNX16AO69A, and 80NSSC21K0316.
