Sep 13, 2023
Version 1
A simple and efficient protocol for generating transgenic hairy roots using Agrobacterium rhizogenes V.1
Peer-reviewed method
- Shaun Ferguson1,
- Nikolaj B. Abel1,
- Dugald Reid2,
- Lene H. Madsen1,
- Thi-Bich Luu1,
- Kasper R. Andersen1,
- Jens Stougaard1,
- Simona Radutoiu1
- 1Department of Molecular Biology and Genetics, Aarhus University, Denmark;
- 2Department of Animal, Plant and Soil Sciences, School of Agriculture, Biomedicine and Environment, La Trobe University, Melbourne, Australia
- PLOS ONE Lab ProtocolsTech. support email: [email protected]

External link: https://doi.org/10.1371/journal.pone.0291680
Protocol Citation: Shaun Ferguson, Nikolaj B. Abel, Dugald Reid, Lene H. Madsen, Thi-Bich Luu, Kasper R. Andersen, Jens Stougaard, Simona Radutoiu 2023. A simple and efficient protocol for generating transgenic hairy roots using Agrobacterium rhizogenes. protocols.io https://dx.doi.org/10.17504/protocols.io.261ge3xkjl47/v1
Manuscript citation:
Ferguson S, Abel NB, Reid D, Madsen LH, Luu TB, et al. (2023) A simple and efficient protocol for generating transgenic hairy roots using Agrobacterium rhizogenes. PLOS ONE 18(11): e0291680. https://doi.org/10.1371/journal.pone.0291680
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 25, 2023
Last Modified: September 13, 2023
Protocol Integer ID: 75854
Keywords: generating transgenic hairy root, using agrobacterium rhizogene, agrobacterium rhizogenes for decade, transgenic hairy root, agrobacterium rhizogene, subsequent transgenic hairy root transformation, causative agent of hairy root disease, rhizobium rhizogene, interkingdom dna delivery tool, limited to pbr322 acceptor strain, efficient construction of recombinant vector, pbr322 acceptor strain, transformation of other plant, hairy root disease, recombinant vector, new modular cloning, effective screening of transconjugant, compatible pbr322, transconjugants from this system, gene, other plant
Funders Acknowledgements:
This work was funded by the project Molecular Mechanisms and Dynamics of Plant–microbe interactions at the Root–Soil Interface (InRoot)
Grant ID: NNF19SA0059362
Abstract
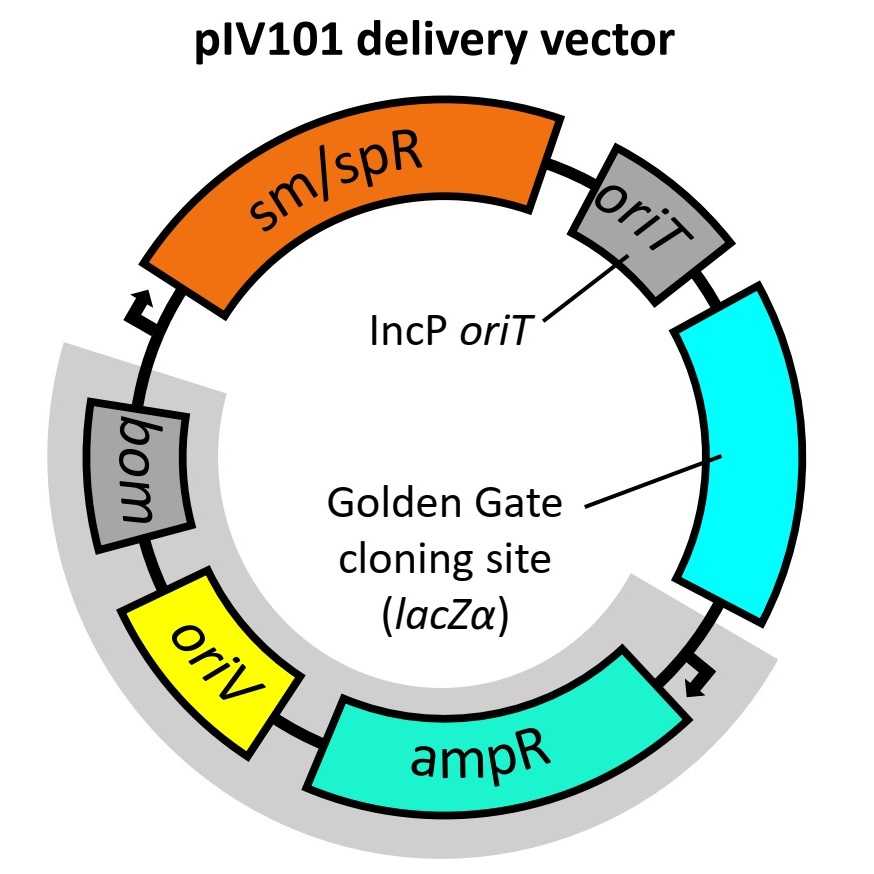
For decades, Agrobacterium rhizogenes (now Rhizobium rhizogenes), the causative agent of hairy root disease, has been harnessed as an interkingdom DNA delivery tool for generating transgenic hairy roots on a wide variety of plants. One of the strategies involves the construction of transconjugant R. rhizogenes by transferring gene(s) of interest into previously constructed R. rhizogenes pBR322 acceptor strains; little has been done, however, to improve upon this system since its implementation. We developed a simplified method utilising bi-parental mating in conjunction with effective counterselection for generating R. rhizogenes transconjugants. Central to this was the construction of a new Modular Cloning (MoClo) compatible pBR322-derived integration vector (pIV101). Although this protocol remains limited to pBR322 acceptor strains, pIV101 facilitated an efficient construction of recombinant vectors, effective screening of transconjugants, and RP4-based mobilisation compatibility that enabled simplified conjugal transfer.Transconjugants from this system were tested on Lotus japonicus and found to be efficient for the transformation of transgenic hairy roots and supported infection of nodules by a rhizobia symbiont. The expedited protocol detailed herein substantially decreased both the time and labour for creating transconjugant R. rhizogenes for the subsequent transgenic hairy root transformation of Lotus, and it could readily be applied for the transformation of other plants.
Guidelines
The Modular Cloning (MoClo) strategy implemented for pIV101 is based on the publication by Weber et al. 2011.
For this protocol we use E. coli strain ST18 for transformation of our cloned construct and subsequent biparental mating to transfer the construct to R. rhizogenes. Another E. coli strain could be substituted but it must contain the RP4 conjugative machinery for mobilisation of pIV101 and we would recommend that it also contains the lacZΔM15 mutation for blue white/screening as well as an auxotrophy for efficicient conterselection (E.g. ΔdapA).
Ensure you wash the cells before and after biparental mating. This ensures adequate removal of both the antibiotics for plasmid selection, and the supplements required by the auxotrophic E. coli.
For standard molecular biology techniques (E.g. PCR, plasmid preparation etc.) please refer to Molecular Cloning: A Laboratory Manual, 4th edition.
Materials
Chemicals for Golden Gate reaction
- Vector backbone (pIV101)
- Additional assembly piece(s)
- 10X NEB BSA (If using BsaI-HF®v2)
- NEB BsaI-HF®v2
- NEB BbsI-HF®
- 10X NEB T4 Ligase Buffer
- NEB T4 Ligase, 2,000,000 cohesive end units/ml
- Nuclease-free water
Chemicals for E. coli chemical transformation
- Filter sterilised diaminopimelic acid (DAP) stock at 30 mM in water (For ΔdapA mutant E. coli strains).
- Previously prepared chemically competent aliquots of E. coli donor strain.
- KCM buffer (5X)
- 5-Bromo-4-chloro-3-indolyl β-D-galactopyranoside (X-gal) 1:1000
Media for bacteria
- LB medium
Material for seed germination and hairy root transformation
- Sand paper
- Hypochlorite (12% stock solution)
- Nuclease-free water
- Filter paper
- Gamborg B5 media including vitamins (Duchefa-biochemie)
- Scalpel for plant cutting
- Syringe needles (0.4 mm)
- Parafilm
- 3M Milipore tape
Antibiotics(Stock)
- Rifampicin (50 mg/ml DMSO)
- Streptomycin (50 mg/ml water)
- Spectinomycin (50 mg/ml water)
- Ampicillin (100 mg/ml water)
Buffers and stock solutions
KCM Buffer (5X):
- 0.5M KCl
- 0.15 M CaCl2
- 0.25 M MgCl2
- Filter sterilise through 0.2 uM filter. Store at -20.
5-Bromo-4-chloro-3-indolyl β-D-galactopyranoside (X-gal):
- 20 mg/ml in dimethylformamide (2% w/v)
Auxotrophic E. coli supplement
5-aminolevulinic acid (ALA):
- 50 mg/ml dissolved in water and filter sterlised
Diaminopemelic acid (DAP):
- 30 mM dissolved in water and filter sterlised
General materials
- Thermocycler suitable for Golden Gate reactions
- Tabletop microcentrifuge
- Spectrophotometer
- 1.5 ml nuclease-free microcentrifuge tubes
- Round and square (11 cm2) petri dishes
Troubleshooting
Safety warnings
Please refer to the manufacturers safety warnings for the individual reagents and chemicals used throughout.
Before start
Ensure you have an appropriate E. coli cloning/donor strain that contains the RP4 conjugative machinery to enable conjugal transfer of pIV101. Some commonly used strains for this include: E. coli S17/ST18, E. coli MFDpir, E. coli WM3064.
If using an auxotrophic E. coli strain such as ST18 or MFDpir make sure you have the required supplement (e.g. 5-aminolevulinic acid (ALA) for ST18 or diaminopimelic acid (DAP) for MFDpir).
Ensure you have a pBR322-derived R. rhizogenes acceptor strain e.g. R. rhizogenes AR1193.
Golden Gate cloning reaction with plasmid pIV101
Add the following components to a Golden Gate reaction:
- Plasmid pIV101 DNA (100 ng ) - X µL
- Insert(s) DNA (100 ng ) - X µL
- Ligase buffer (10x) - 1 µL
- T4 DNA Ligase - 1 µL
- BpiI (BbsI) (or BsaI for level 1 constructs) - 1 µL
- MilliQ water - up to final volume of 10 µL
15m
Incubate reaction in thermocycler with the following steps:
| Temperature | Time | Cycles | |
| 37C | 00:05:00 | 30-60 X | |
| 16C | 00:05:00 | ||
| 80C | 00:10:00 | 1 |
Note
If the Golden Gate reaction is failing to incorporate your desired insert, an additional step that may help:
Following completion of the reaction cycle from step 1.1 - add 1 μl of the restriction enzyme used in the reaction (step 1) to the mix again and incubate for a further 1 - 2hrs at 37C
Because the ligase has already been deactivated, additional active restriction enzyme can help to deplete any remaining plasmids that do not contain the desired insert fragments
4h
Transformation of chemical competent E. coli ST18
Mix in a 1.5 ml tube:
- 20 µL 5X KCM buffer
- 5 µL Golden Gate cloning mix (from step 1.1 following completion of the program)
- 75 µL ddH2O
5m
Incubate for 00:02:00 on ice
2m
Thaw a 100 µL aliquot of chemically competent E. coli ST18 cells on ice
Mix together the chemical competent E. coli cells and the reaction mixture from step 2 by pipetting
1m
Incubate the mix for 00:20:00 on ice
20m
Incubate for 00:10:00 at Room temperature
10m
Add 800 µL LB with 5 μg/ml 5-aminolevulinic acid (ALA) and grow for 01:00:00 at 37 °C with shaking
1h
Centrifuge to pellet the cells at 14000 rcf, 25°C, 00:02:00
2m
Resuspend the pellet in 100 µL of LB containing 50 μg/ml ALA
2m
Plate out the 100 µL of resuspended pellet onto LB agar containing 100 μg/ml ampicillin, 150 μg/ml spectinomycin, and 50 μg/ml ALA
5m
- If using blue/white selection then also add 2% (w/v) 5-Bromo-4-chloro-3-indolyl β-D-galactopyranoside (X-gal) to the media
Grow at 37 °C Overnight and check for colonies the following day
Note
If you are having difficulties obtaining E. coli transformants than we suggest trying electrocompetent E. coli cells which have a higher transformation efficiency. For transformation by electroporation we recommend first performing a clean up of the Golden Gate reaction mixture DNA to prevent arcing
16h
If using X-gal for selection avoid blue colonies as pIV101 contains the lacZα fragment in the GGA cloning site
In parallel with the next step (step 13), select colonies from the transformation (step 12) and confirm the construct from the Golden Gate assembly. This can be carried out by several standard approaches:
- Perform colony PCR to amplify the region of the plasmid that contains the GGA cloning site to ensure that the cloning site contains the expected insert size
- This product can be sent for further confirmation by Sanger sequencing
- Additionally, perform a plasmid preparation (miniprep) from the E. coli clone which can then be used as template for PCR amplification or for whole plasmid sequencing
Note
If your insert is large then we recommended sending the construct for whole plasmid sequencing as an alternative to confirmation by standard Sanger sequencing
1d
R. rhizogenes transformation
Note
This section and the next section can be carried out in parallel to save time
Start liquid culture of the wild-type Agrobacterium rhizogenes (now Rhizobium rhizogenes) from a single colony in LB media with 100 μg/ml rifampicin (a 5 ml broth is sufficient)
10m
Incubate for 48:00:00 at 28 °C with shaking
2d
Inoculate an LB broth containing 100 μg/ml ampicillin, 150 μg/ml spectinomycin, and 50 μg/ml ALA with a single colony for an ST18 clone carrying the construct of interest (from the previous section)
5m
Incubate the LB broth from step 15 Overnight at 37 °C with shaking.
1d
Centrifuge 1 ml of the overnight broth for the E. coli ST18 culture carrying the construct of interest at 14000 rcf, 00:02:00
2m
Resuspend the E. coli pellet in 1 mL sterile dH2O and repeat the previous step. This is to wash away the broth culture containing antibiotics
3m
Resuspend the E. coli pellet in 50 µL sterile dH2O
2m
Centrifuge 1 ml of the broth culture of R. rhizogenes from step 13-14 at 8000 rcf, 00:02:00
2m
Resuspend the R. rhizogenes pellet in 1 mL sterile dH2O and repeat the previous step. This is to wash away the broth culture containing antibiotics
3m
Resuspend the R. rhizogenes pellet in 50 µL sterile dH2O
2m
Perform a biparental mating by mixing the resuspended E.coli ST18 (step 19) and R. rhizogenes (step 22) in 100 µL total volume and spot onto plates of LB media supplemented with 50 μg/ml ALA (but no antibiotics) and then wait until the spot is dry
30m
Grow the biparental mating spot plates Overnight at 28 °C
16h
Scrape the biparental mating spot of the E. coli ST18 clone + R. rhizogenes and resuspend in 1 mL sterile dH2O
5m
Centrifuge at 14000 rcf, 00:02:00 to pellet, and resuspend in 1 mL sterile dH2O. This step should wash away any residual supplement from the mating plates that enables E. coli growth
2m
Centrifuge again at 14000 rcf, 00:02:00 to pellet, and resuspend in 100 µL sterile dH2O (The total volume will be more due to the pellet)
2m
Transfer the resuspended mix from the previous step onto LB media plates supplemented with 100 μg/ml ampicillin, 50 μg/ml spectinomycin, and 100 μg/ml rifampicin. (No ALA). Plate out for single colonies
Note
For step 28: if the bacterial suspension is too thick then try diluting the resuspended mix by 1:10 in sterile water and plate this out in parallel
2m
Incubate the plate(s) for 48:00:00 at 28 °C
2d
Re-streak the R. rhizogenes strains carrying the construct of interest on LB agar containing 100 μg/ml ampicillin, 50 μg/ml spectinomycin, and 100 μg/ml rifampicin to ensure single colonies
5m
Incubate the plate(s) for 48:00:00 at 28 °C
2d
Preparation of seeds for hairy root transformation (for Lotus japonicus)
To scarify the seeds, transfer the required number of seeds to a mortar and rub them with sand paper until they become white on the ends
2m
Transfer the seeds to a sterile tube (at least 15 ml capacity) and sterilise the seeds by immersing them in a 1% hypochlorite solution, and incubate in this solution for 15 min at Room temperature
15m
Remove the hypochlorite solution and discard appropriately. Add sterile water and invert the tube several times. Repeat this 5 times to wash the seeds and remove any residual hypochlorite
5m
Fill the tube with sterile water and incubate for at least 02:00:00 at Room temperature with shaking (alternatively incubate Overnight at 4 °C )
2h
Using sterile forceps, transfer seeds to square petri dishes containing sterile filter paper soaked in sterile dH2O (approximately 15 min per plate)
15m
Incubate the square plates containing the surface sterilised seeds sitting on damp filter paper
from the previous step for 72:00:00 at 21 °C
3d
Transfer germinated seeds to a square petri dish that contains solid Gamborg B5 media including vitamins
15m
Grow seedlings for 72:00:00 at 21 °C with 16H/8H light/dark cycle, until the root has attached itself to the media
3d
Preparation of R. rhizogenes for hairy root transformation
Resuspend the R. rhizogenes strain from the agar plate (step 30-31) into sterile dH2O as a thick suspension (OD600 >2). Approximately 100 µL is needed per plant, so make sure the volume of water for the suspension is sufficient for the number of plants that will be transformed
Note
The actual concentration is not important, there just needs to be an excess of the R. rhizogenes
10m
Hairy root transformation of Lotus seedlings with R. rhizogenes
Wound seedlings with syringe needle (0.4mm) at the hypocotyl
1m
Add one large drop (~ 100 µL ) of the thick suspension of R. rhizogenes (from step 40) on top of the wound
1m
Incubate the seedling for 01:00:00 horizontally to let the R. rhizogenes infect the hypocotyl
1h
Seal the plates containing the now infected seedlings with parafilm on the sides and bottom (prevents dehydration). Seal top with micropore tape on the top edge (Keeps the plates sealed but allows gas exchange)
1m
Grow the infected seedlings for 72:00:00 at 21 °C in the dark to enhance infection
3d
Grow the infected seedlings for 3 weeks at 21 °C with 16H/8H light/dark cycle until transformed roots emerge
3w
Selecting hairy roots
Hairy roots will emerge and develop from the infected wound sites
Place the plates containing the transformed plants on a transilluminator. Remove the non-fluorescently labelled roots (untransformed roots) using a scalpel
Note
The Golden Gate assembly constructs should be designed to include a fluorescent marker to distinguish hairy roots from non-transformed roots. Alternative markers like RUBY can also be used
1h
Transfer plants with transformed roots to new pots or plates and grow plants at 21 °C with 16H/8H light/dark cycle for one week
1w
For rhizobium inoculation of hairy roots (optional)
Resuspend the rhizobium strain (or your bacteria of interest) from a freshly streaked agar plate into sterile dH2O
10m
Adjust the OD600 of the suspension to between 0.01 - 0.05 in a volume that is sufficient to provide 100 µL per plant to be inoculated
15m
If plants are on square agar plates: lay the square plate flat. Inoculate the roots of the plants by carefully applying 100 µL of the suspension from the previous step using a pipette. Ensure that you apply the inoculum evenly to as much of the root as possible
5m
Leave inoculated plant plate(s) flat for a short period so that the bacterial suspension can spread evenly across the plate and ensure contact with the roots
5m
If the plants are in pots: determine the volume that is equal to 100 µL X the number of plants present, and distribute this evenly to the plant pot substrate using a pipette
5m
Grow the inoculated plants for 3 weeks post inoculation at 21 °C with 16H/8H light/dark cycle until nodules are formed
3w
Protocol references
Golden Gate cloning:
Engler C, Kandzia R, Marillonnet S. A one pot, one step, precision cloning method with high throughput capability. PloS One. 2008;3(11):e3647. doi: https://doi.org/10.1371/journal.pone.0003647.
Modular Cloning (MoClo):
Weber E, Engler C, Gruetzner R, Werner S, Marillonnet S. A modular cloning system for standardized assembly of multigene constructs. PloS One. 2011;6(2):e16765. doi: https://doi.org/10.1371/journal.pone.0016765.
E .coli strains:
Ferrieres L, Hémery G, Nham T, Guérout A-M, Mazel D, Beloin C, et al. Silent mischief: bacteriophage Mu insertions contaminate products of Escherichia coli random mutagenesis performed using suicidal transposon delivery plasmids mobilized by broad-host-range RP4 conjugative machinery. Journal ofBacteriology. 2010;192(24):6418-27. doi: https://doi.org/10.1128/JB.00621-10.
Thoma S, Schobert M. An improved Escherichia coli donor strain for diparental mating. FEMS Microbiology Letters. 2009;294(2):127-32. doi: https://doi.org/10.1111/j.1574-6968.2009.01556.x.
R. rhizogenes pBR322-acceptor strains:
Stougaard J, Abildsten D, Marcker KA. The Agrobacterium rhizogenes pRi TL-DNA segment as a gene vector system for transformation of plants. Molecular and General Genetics MGG. 1987;207(2):251-5. doi: https://doi.org/10.1007/BF00331586.
Radutoiu S, Madsen LH, Madsen EB, Nielsen AM, Stougaard J. Agrobacterium rhizogenes pRi TL-DNA integration system: a gene vector for Lotus japonicus transformation.Lotus japonicus Handbook: Springer; 2005. p. 285-7.
Green, M. R., & Sambrook, J. (2012). Molecular cloning. A Laboratory Manual 4th.
