Feb 17, 2026
A 4-5X expansion microscopy protocol for high‑resolution imaging of olfactory sensory neuron cilia in mouse olfactory epithelium
- Ebsy Jaimon1,
- Suzanne Pfeffer1
- 1Stanford University School of Medicine and Aligning Science Across Parkinson's

Protocol Citation: Ebsy Jaimon, Suzanne Pfeffer 2026. A 4-5X expansion microscopy protocol for high‑resolution imaging of olfactory sensory neuron cilia in mouse olfactory epithelium. protocols.io https://dx.doi.org/10.17504/protocols.io.6qpvry22ogmk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 16, 2026
Last Modified: February 17, 2026
Protocol Integer ID: 243410
Keywords: Olfactory epithelium, olfactory sensory neurons, cilia, imaging of olfactory sensory neuron cilia, resolution imaging of olfactory sensory neuron, olfactory sensory neuron cilia, mouse olfactory epithelium, olfactory sensory neuron, 5x expansion microscopy protocol, using conventional confocal microscopy, delicate ciliary structures with hydrogel, delicate ciliary structure, expansion microscopy, conventional confocal microscopy, microscopy protocol, ciliary architecture, rapid dissection of the o, resolution imaging
Funders Acknowledgements:
Aligning Science Across Parkinson's
Grant ID: ASAP-000463
Abstract
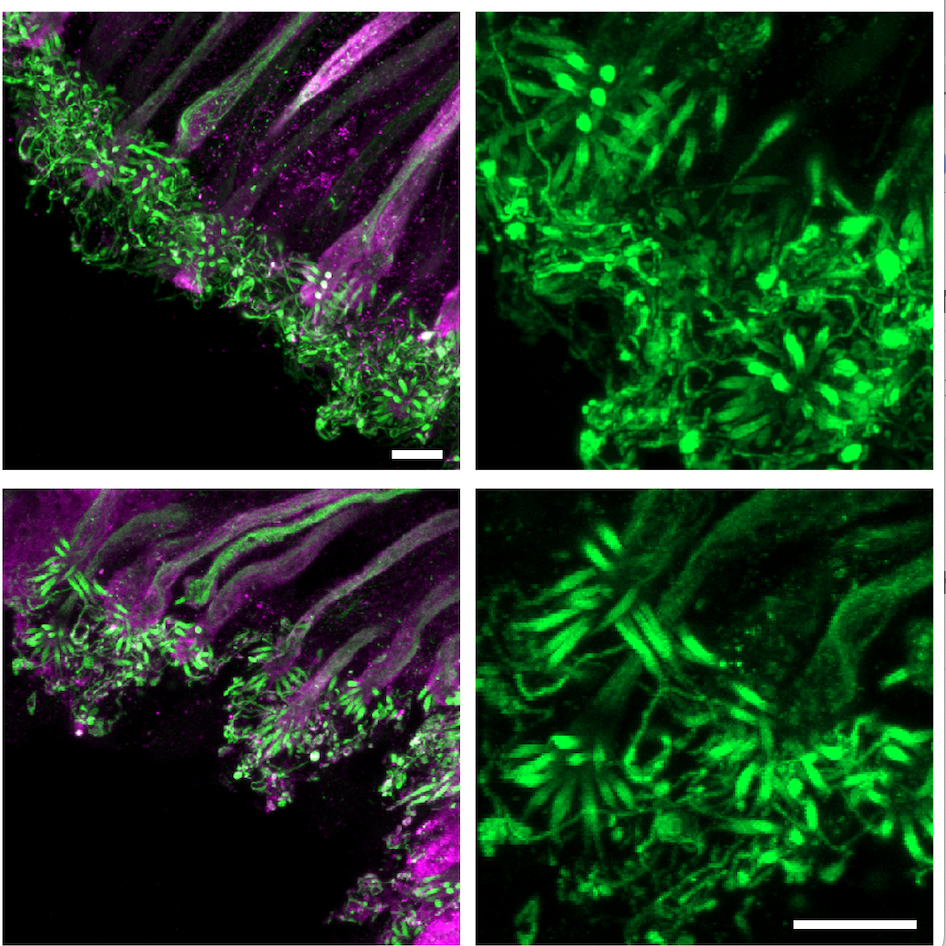
Here, we present a protocol for 4-5× expansion of mouse olfactory epithelium (OE), optimized for high-resolution imaging of olfactory sensory neuron (OSN) cilia. The method combines rapid dissection of the OE on a cold platform to preserve delicate ciliary structures with hydrogel embedding and expansion microscopy (ExM) to achieve isotropic 4-5 fold expansion (based on references 2-4). This approach enables detailed visualization of OSN ciliary architecture using conventional confocal microscopy, without the need for specialized super-resolution instrumentation. OSNs were labeled using anti-Olfactory Marker Protein (OMP) and their cilia were labeled using anti-acetyl-tubulin antibodies.
Materials
Materials:
- Wild-type C57BL/6J mice (Jackson Laboratory #:000664)
- 25G X 1 ½ needle (BD # 305127)
- 10ml syringe ( BD #302995)
- Isoflurane (MWI Veterinary Supply Co # 13985-528-60)
- Paraformaldehyde (PFA) 32% solution (Electron Microscopy Sciences #15714-S)
- Leica Dissecting microscope MZFLIII
- Bone Nipper (F.S.T # 16102-11)
- Bonn Miniature Iris Scissors (Miltex # 18-1392)
- Micro Friedmann-Pearson Rongeur (F.S.T # 16220-14)
- Spring scissors curved (Roboz # RS-5675)
- #2 Forceps (F.S.T # 11223-20)
- Sucrose (Millipore # 8510)
- 22 X 22 X 20 mm Embedding mold (epredia #2219)
- Tissue-Tek O.C.T compound (Sakura #4583)
- Superfrost plus slides 25 X 75 X 1 mm (VWR # 48311-703)
- PAP pen (Vector Laboratories # H-4000)
- Coverslips 22 X 22 mm No. 1.5 (VWR # 48366-227)
- Acrylamide 98+%(Alfa Aesar # A17157)
- Bis-acrylamide >99.9% (VWR # 0172)
- Sodium acrylate 97% (Sigma Aldrich # 408220)
- Ammonium persulfate (APS) ≥98%(Sigma Aldrich # A7460)
- TEMED (Fischer Bioreagents #BP150-100)
- Parafilm M sealing film (# HS234526B)
- SDS (CalbioChem # 7910)
- Tris base (Chemcruz # sc-3715)
- Sodium chloride (NaCl) (J.T Baker # 3624-01)
- Bovine serum albumin (BSA) (GoldBio #A-420)
- Triton-X-100 (Sigma Aldrich # T8787)
- Goat polyclonal anti-OMP (FUJIFILM Wako Pure Chemical Corporation (#54-10001-WAKO)
- Mouse monoclonal anti-Acetyl tubulin (Sigma Aldrich # T6793)
- H+L Donkey anti-goat AF 568 (Life Technologies # A11057)
- H+L Donkey anti-mouse AF 488 (Life Technologies #A32766)
- DAPI (Sigma # D9542)
- Poly-D-lysine (Sigma # P6407)
- 6 well glass bottom imaging plates (Cellvis #P06-1.5H-N)
- Zeiss LSM 900 microscope
[All procedures were approved by the institutional animal care and use committee (IACUC)]
Safety note: Acrylamide, sodium acrylate, TEMED, APS, SDS are toxic/irritants and should be handled in a fume hood with PPE, with waste disposed appropriately.
Troubleshooting
Safety warnings
Safety note: Acrylamide, sodium acrylate, TEMED, APS, SDS are toxic/irritants and should be handled in a fume hood with PPE, with waste disposed appropriately.
Ethics statement
All procedures were approved by the institutional animal care and use committee (IACUC).
Dissection of olfactory epithelium and embedding in O.C.T.
Anesthetize the mouse deeply with isoflurane. Ensure there are no reflexes by pinching the toes.
Lay the mouse on a dissecting board and cut through the skin to open the thoracic cavity, exposing the heart.
Identify the left ventricle and right atrium. Insert a 10ml syringe with a 25G needle filled with 10 ml chilled 1X PBS, pH 7.4 into the left ventricle. Make a small cut on the right atrium and perfuse the animal under a continuous slow steady pressure (~1ml/min) to flush blood from the vessels until the liver and extremities become pale.
Switch to 8ml, chilled freshly prepared 4% PFA in 1X PBS, pH 7.4. Perfusion is complete when the body stiffens, and the tissues appear fixed.
Dissect olfactory epithelium (septum and turbinates) rapidly as described by Belfort et al. (2025) under a dissecting microscope. Briefly, after removal of surrounding soft tissue and cranial vault, the brain and nasal bones are excised, and the intact OE is gently released using forceps. Note: Place the tissue on a cold platform (pre-cooled on ice) during dissection to preserve delicate cilia and tissue morphology. The time from perfusion to OE immersion in 4% PFA should ideally be <10–15 minutes.
Post-fix olfactory epithelium in 5ml of 4% PFA in 1X PBS (sufficient volume to fully submerge tissue) at 4°C overnight (up to 24 hours). Rinse the olfactory epithelium in 1X PBS three times (5ml each), for 10-15 minutes each, to remove the fixative.
Cryoprotect the tissues by immersing them in 5ml of 30% sucrose in 1X PBS (sufficient volume to fully submerge tissue) and incubate at 4°C until the tissue sinks to the bottom of the tube. Note: Tissue typically sinks within 4–24 h; do not embed before complete sinking to avoid ice‑crystal artifacts.
Add a small amount of O.C.T. to the bottom of the embedding mold and place it on dry ice and allow it to freeze. This creates a stable base layer.
Next, fill the mold fully with O.C.T. compound. Rinse the tissue quickly in PBS to remove sucrose and place the sample in O.C.T. in the desired orientation. Note the orientation for future reference.
Freeze rapidly by placing the mold back on dry ice.
Store embedded tissues at -80 °C until sectioning.
Cryosection at 14µm thickness using a cryostat set at −18 to −22°C and collect sections on a charged Superfrost slide.
Anchoring and Gelation of OE sections
Prepare 10ml of Monomer Fixative solution
Monomer fixative is used to introduce acrylamide handles and retain proteins
within the gel during expansion.
Transfer the slides with the OE section from -80°C. Let the slides thaw at room temperature for 1-2 minutes. Gently blot any moisture using a Kimwipe, if needed, and then draw a barrier around the tissue using a PAP pen. Let the section air dry for 5 minutes at room temperature.
Gently add 300µl (enough to fully cover the section) of 1X PBS onto the section and wash three times for 5 minutes each.
Remove the 1X PBS and add 300µl (enough to fully cover the section) of monomer fixative within the PAP border. Incubate for 2 hours at RT. Note: Make sure that the solution doesn’t dry out.
During the incubation with monomer fixative, start preparing the following:
17.1 Prepare a humid gelation chamber (doi 10.17504/protocols.io.dm6gp15w1gzp/v1). Use a 1ml pipette tip box with water added at the bottom and Parafilm placed on the tip-holding-insert. Create spacers by stacking five #1.5 coverglasses carefully to create ~1 mm spacer height and place on the Parafilm so that the slide can rest on top of these spacers and the gel will have some room to spread. Pre-cool the gelation chamber at 4°C.
17.2 Prepare 38% w/v sodium acrylate, 50% w/v acrylamide, and 2% w/v bis-acrylamide in water. These stock solutions are required to prepare a master monomer solution.
17.2.1 Prepare 38% w/v sodium acrylate stock by weighing 0.76g of sodium acrylate and adding 1.5 ml of water to dissolve. Mix thoroughly until dissolved. Bring the volume up to 2ml. Store at 4°C. Note: Sodium acrylate should be fresh and of high quality.
17.2.2 Prepare 50% w/v Acrylamide stock by weighing 1g acrylamide and adding 1.5 ml of water to dissolve. Mix thoroughly or vortex until dissolved. Bring the volume to 2ml. Store at 4°C protected from light.
17.2.3 Prepare 2% w/v Bis acrylamide stock by weighing 0.1 g bis acrylamide and adding 4ml of water. Mix until completely dissolved. Bring volume to 5 ml with water. Store at 4°C.
17.3 Prepare 4ml, master monomer solution from the stock solutions prepared in the previous step. Store the remaining aliquots at -20°C. Use within 2 weeks of preparation.
Note: The master monomer solution is without TEMED and APS.
17.4 Prepare 10% TEMED in deionized water and keep it on ice.
17.5 Prepare 10% APS in deionized water by weighing 0.2g of APS and adding 2 ml of water to dissolve. Keep on ice.
Note: 10% APS and master monomer solution can also be prepared ahead and stored at -20°C. Thaw the master monomer solution and 10% APS on ice if stored at -20°C or keep the prepared master monomer solution on ice until use. Avoid repeated freeze–thaw cycles.
For each section, aliquot 44 µl of master monomer solution on ice. Critical: Steps 19–21 must be performed rapidly to prevent premature gelation.
Remove monomer fixative solution from the section and add master monomer solution (without APS and TEMED) to wash the sections. Incubate at RT for 2-3 minutes.
Next, transfer the slide to a cold room or a cold metal block and remove the master monomer solution from the section.
Immediately before gelation, add 2.75 µl, 10% TEMED and 2.75 µl, 10% APS (5.6% of total volume each) to each master monomer aliquot, mix briefly and pipette (~50 µl per section) onto the Parafilm between the spacers. Quickly invert the slide containing the section and position the sample over the droplet. Add a small weight (e.g., glass slide or small metal nut) to achieve even spreading.
Note: Once APS/TEMED have been added, polymerization is time‑ and temperature‑dependent; process slides rapidly or work in small batches on ice to maintain uniform gelation.
Incubate the gel at 4°C for 5 minutes to initiate gelation.
Continue incubating the gel in a humid chamber at RT for 2 hours or overnight.
Denaturation and expansion
Prepare 200ml of denaturation buffer.
Note: Dissolve solutes in 150ml water. Adjust pH to 9, then bring up to the final volume.
Invert the slide and gently peel off the Parafilm from the gel. Trim excess gel from regions without the sample if needed.
Photograph the gel and measure the size of the gel using a ruler to estimate the later expansion factor.
Transfer the slide with gel to a wide glass container containing sufficient denaturation buffer to fully submerge the gel (typically 100–200 ml) and incubate for 6 hours at 95°C on a dry heat block, covering the container with aluminum foil to minimize evaporation.
After 6 hours of incubation, the gel will now be off the slide. Note: Over‑denaturation (longer than 6–8 h at 95 °C) can cause gel fragility or epitope loss and under‑denaturation can reduce expansion/isotropy.
Begin expansion by transferring the gel into a fresh beaker or a wide dish filled with 50-100 ml ddH20 and incubating for 30 minutes at RT with gentle orbital shaking. Note: Use a wide spatula or paintbrush to handle gels. Use care with forceps to prevent tearing.
Repeat step #29 again by incubating in 50-100 ml ddH20 for 30 minutes at room temperature with gentle orbital shaking.
Repeat once again for 30 minutes at room temperature, or let the gel sit overnight at room temperature with gentle orbital shaking.
Measure expansion factor using a ruler. Note: For initial optimization, measure expansion after each water change to confirm when the gel reaches its plateau expansion.
Figure 1: Image of gels before (left) and after (right) expansion. The beige circles are the tip-container holes; the clear gel (before and after) is circled with white dashed lines.
Transfer the gel to a fresh beaker or wide dish with 50-100ml PBS (Add sufficient volume to allow free movement) and incubate for 30 minutes at room temperature with gentle orbital shaking.
Note: The gels shrink a bit in PBS but not back to their original size.
Primary antibody incubation
Prepare the primary antibody dilution as shown below.
Note: Triton X-100 is included during primary antibody but not secondary incubation, as permeabilization is achieved during denaturation. No separate blocking step is used; blocking is achieved by including 3% BSA in the primary and secondary antibody diluents.
Transfer the gel into a 2ml tube with ~1.5-2 ml diluted primary antibody and incubate at 4°C overnight in a nutator. Note: The gel should be fully submerged and able to move freely in the tube; if the gel is larger, switch to a 15 ml conical tube and scale the antibody volume so these conditions are still met. Use a volume that still completely covers the gel when it is lying flat and has some room to move during nutation.
Secondary antibody incubation
Transfer the gel to a fresh beaker or wide dish containing 50-100ml PBS and incubate for 30 minutes at room temperature with gentle orbital shaking.
Wash once again in 50-100ml PBS and incubate for 30 minutes at room temperature with gentle orbital shaking.
Prepare the secondary antibody dilution.
Transfer the gel into a 2ml tube with ~1.5-2ml diluted secondary antibody and incubate at 4°C overnight in a nutator. Note: The gel should be fully submerged and able to move freely in the tube.
Final Expansion and Imaging
Transfer the gel to a beaker or wide dish with 50-100ml PBS and incubate for 30 minutes at room temperature with gentle orbital shaking .
Wash once again in 50-100ml PBS and incubate for 45 minutes at room temperature with gentle orbital shaking.
Place gel in approximately 50–100 ml ddH₂O (sufficient to fully immerse and allow free expansion of the gel) and incubate at room temperature for 30 minutes with gentle orbital shaking.
Repeat again in approximately 50-100ml ddH20 and incubate at room temperature for 30 minutes with gentle orbital shaking.
Allow the gels to fully expand in 50-100ml ddH20 for 1 hour. Measure the expansion factor again using a ruler.
During the final expansion, coat the wells of the imaging plates with 0.1 mg/ml poly-D-lysine in sterile water. Incubate for 20 minutes at room temperature. Remove the solution and rinse gently with water. Air dry at room temperature. Store at 4°C or use the same day.
Gently blot the gel with a Kimwipe to absorb excess water.
Use an uncoated dish to ensure that the sample side (side that was originally in contact with the slide bearing OE sections) of the gel is placed towards the objective. Then, place the gel on the coated dish and lightly press the gel using a paintbrush to remove bubbles, and use a Kimwipe to absorb excess water. Note: Use minimal residual water when placing the gel on the coated surface to maximize adhesion; excess water can allow gel drift during imaging. Blot edge with Kimwipe as needed.
Image using Zeiss LSM 900. Preview the sample at a lower magnification(5X and 20X) and once the region of interest is in focus, shift to higher magnification(63X oil immersion objective). The recommended z-step size is 0.3-0.4µm.
Figure 2: Olfactory sensory neurons stained for cilia (acetylated tubulin) and cell type marker (anti-OMP); scale bars = 2µm after expansion. These neurons are multi-ciliated with ~10-30 cilia per neuron.
Protocol references
- Belfort, B. D. W., Insalaco, A. M., Chew, C. C., Jia, J. D., Younis, J., & Arenkiel, B. R. Rapid dissection and dissociation of the mouse olfactory epithelium for single-nucleus suspensions. Journal of Visualized Experiments (222), e68472 (2025).
- Chen, F., Tillberg, P. W., & Boyden, E. S. Expansion microscopy. Science 347(6221), 543–548 (2015).
- Wassie, A. T., Zhao, Y., & Boyden, E. S. Expansion microscopy: principles and uses in biological research. Nature Methods 16, 33–41 (2019).
- Ching, K., Wang, J. T., & Stearns, T. Long-range migration of centrioles to the apical surface of the olfactory epithelium. eLife 11, e74399 (2022).
