Sep 03, 2025
2-step PCR mixture and conditions (Barcoded-head primers for seqs pooling)
- Yin-Tse Huang1,
- Tsu-Chun Hung1
- 1Kaohsiung Medical University

Protocol Citation: Yin-Tse Huang, Tsu-Chun Hung 2025. 2-step PCR mixture and conditions (Barcoded-head primers for seqs pooling). protocols.io https://dx.doi.org/10.17504/protocols.io.n92ld6rd7g5b/v1
Manuscript citation:
Herbold CW, Pelikan C, Kuzyk O, Hausmann B, Angel R, Berry D, Loy A. 2015. A flexible and economical barcoding approach for highly multiplexed amplicon sequencing of diverse target genes. Front. Microbiol. [Internet] 6:731. Available from: http://dx.doi.org/10.3389/fmicb.2015.00731
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 02, 2025
Last Modified: September 03, 2025
Protocol Integer ID: 226283
Keywords: pcr mixture, step pcr mixture, 2x supergreen pcr master mix, head primers for seq, pcr, head primer, primer, mix, seq
Abstract
PCR mixture and condition (2X SUPERGREEN PCR MASTER MIX)
Troubleshooting
Wear glove, clean up the working bench w. 1% bleach
For 1' PCR head-primers
Prepare 1' PCR master mixutre for head-primers (prepare 1.2X of solutions for pipetting error if needed)
PCR mixture for head-primers for each reaction
| A | B | C | D | |
| Component | Volume | Volume (1.2X) | Final conc. | |
| PowerPol 2X PCR Master Mix | 12.5 μL | 15 μL | 1X | |
| Forward Primer (10 µM) | 0.5 μL | 1.2 μL | 0.2 µM | |
| Reverse Primer (10 µM) | 0.5 μL | 1.2 μL | 0.2 µM | |
| ddH2O | 10.5 μL | 11.1 μL | - | |
| Total volume | 24 μL | 28.5 μL |
| A | B | C | D | |
| Component | Volume | Volume (1.2X) | Final conc. | |
| 2X KAPA HiFi HotStart ReadyMix | 12.5 µL | 15 μL | 1X | |
| Forward Primer (10 µM) | 0.75 µL | 0.9 µL | 0.3 μM | |
| Forward Primer (10 µM) | 0.75 µL | 0.9 µL | 0.3 μM | |
| ddH2O | 10 μL | 12 µL | - | |
| Total volume | 24 μL | 28.8 μL | - |
Note
Negative control ALWAYS NEEDED! For example, if you have 5 PCR reactions to run, prepare master mixture for 6 reactions (5 DNA template + 1 negative control).
Mix the 1' PCR master mixture gently by pippeting. Quick spin the tube.
Transfer 24 µL 1' PCR master mixutre in 8-strip PCR tubes.
Add 1 µL DNA template in 8-strip PCR tubes, resulting in a 25 µL reaction mixture for 1' PCR.
Note
Negative control contains only 24 µL master mixture but not DNA template
Mix the reaction mixture gently by tapping the tubes. Quick spin the tubes.
Carry out PCR using the following condition:
1' PCR condition for head-primers
| A | B | C | D | |
| Step | Temp | Sec | Cycle | |
| Initial denaturation | 95 ºC | 180 | ||
| Denaturation | 95 ºC | 30 | 25 cycles | |
| Annealing | 60-66 ºC varied (b) | 30 | ||
| Extension | 72 ºC | 180 | ||
| Final extension | 72 ºC | 420 | ||
| Preservation | Preservation | 4 ºC | ∞ |
b. Annealing varied, 60-66C is working; Refer to 1' PCR primers for annealing temperature
c. 1kb ~ 1min extension; enough time allow full extension of sequence
Carry out electrophoresis for inspection of DNA products
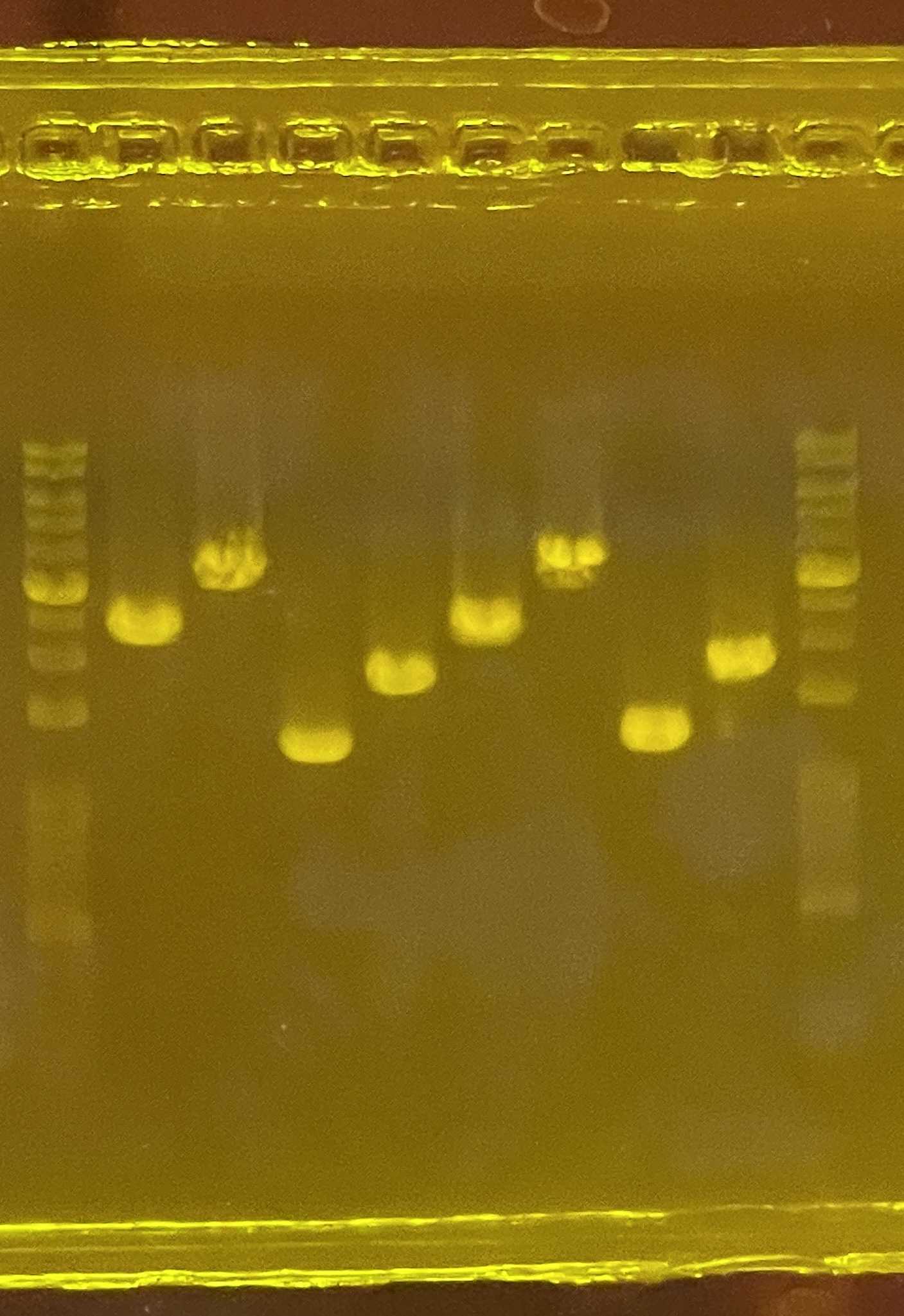
Gel before markdown
Markdown wells and upload the pictures to the Lab Google drive
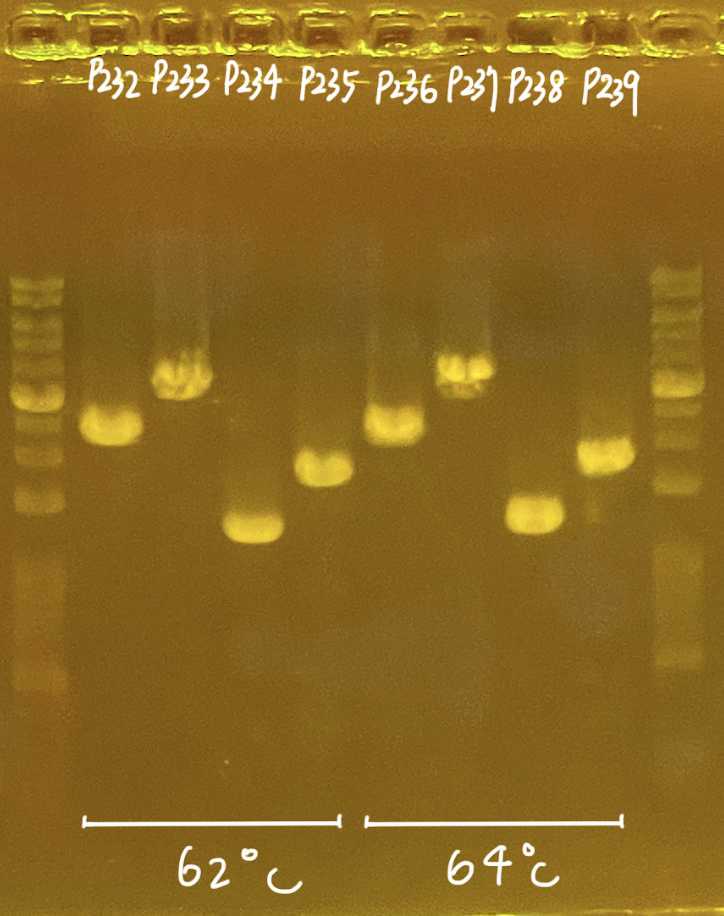
Marked gel picture go to the Lab Google drive
For 2' PCR barcoded-head primers
Prepare 2' PCR master mixutre for barcoded-primers (prepare 1.2X of solutions for pipetting error if needed)
PCR mixture for barcoded-primers for each reaction (NO PRIMERs at this point!!)
| A | B | C | D | |
| Component | Volume | Volume (1.2X) | Final conc. | |
| ZEJU PCR Master Mix | 7.5 µL | 9 µL | - | |
| ddH20 | 5.55 µL | 6.66 µL | - | |
| Total volume | 13.05 µL | 15.66 µL | - |
Note
Negative control ALWAYS NEEDED! For example, if you have 5 PCR reactions to run, prepare master mixture for 6 reactions (5 DNA template + 1 negative control).
Mix the 2' PCR master mixture gently by pippeting. Quick spin the tube.
Transfer 13.05 µL of the 2' PCR master mixture to 8-strip PCR tubes.
Add 1.2 µL pre-mixed barcoded-head primers (Forward + Reverse) to each PCR tubes.
Add 0.75 µL of 1' PCR product as template, resulting in 15 µL reaction mixture for 2' PCR.
Negative control contains only 14.25 µL master mixture and premixed barcoded-head primers but not DNA template
Mix gently by tapping the tubes. Quick spin the tubes.
Carry out 2' PCR using the following condition:
2' PCR condition for barcoded-head primers
| A | B | C | D | |
| Step | Temp | Sec | Cycle | |
| Initial denaturation | 98 ºC | 30 | ||
| Denaturation | 98 ºC | 15 | 12 cycles | |
| Annealing | 64-68 ºC varied (a) | 15 | ||
| Extension | 72 ºC | 20 (b) | ||
| Final extension | 72 ºC | 210 | ||
| Preservation | Preservation | 4 ºC | ∞ |
a. Annealing varied, 65 C is working based on test on 220531; Refer 2' PCR primers for annealing temperature
b. 1kb ~ 1min extension; enough time allow full extension of sequence
Carry out electrophoresis for inspection of DNA products
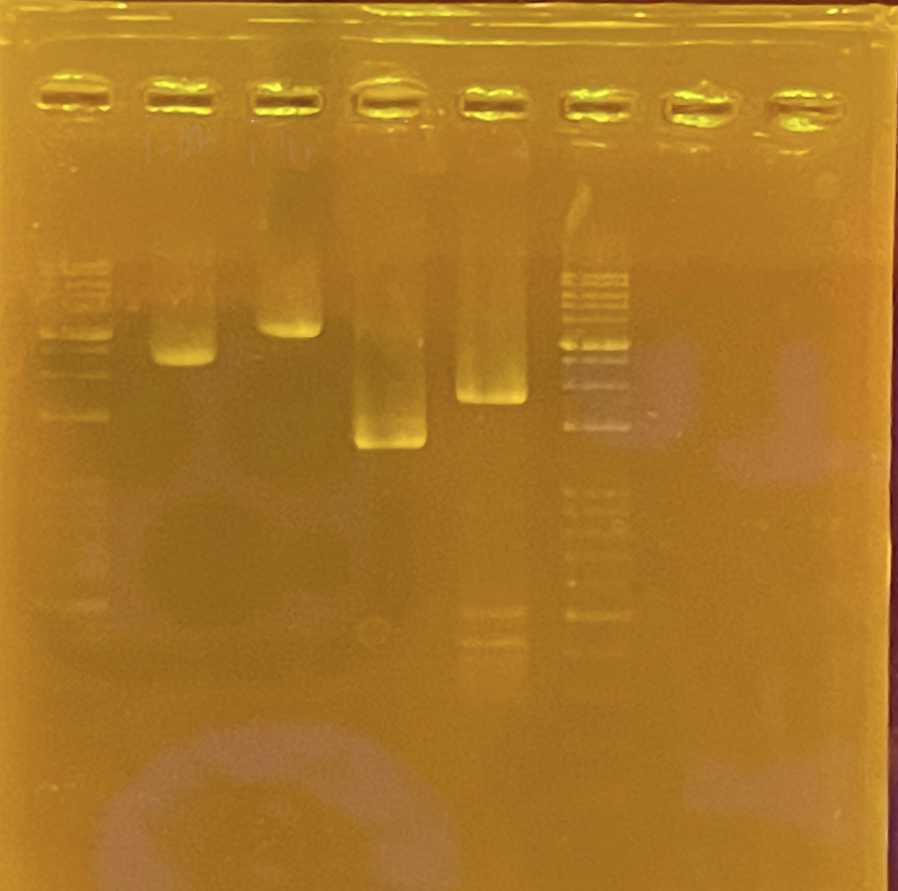
Gel before markdown
Markdown wells and upload the pictures to the Lab Google drive
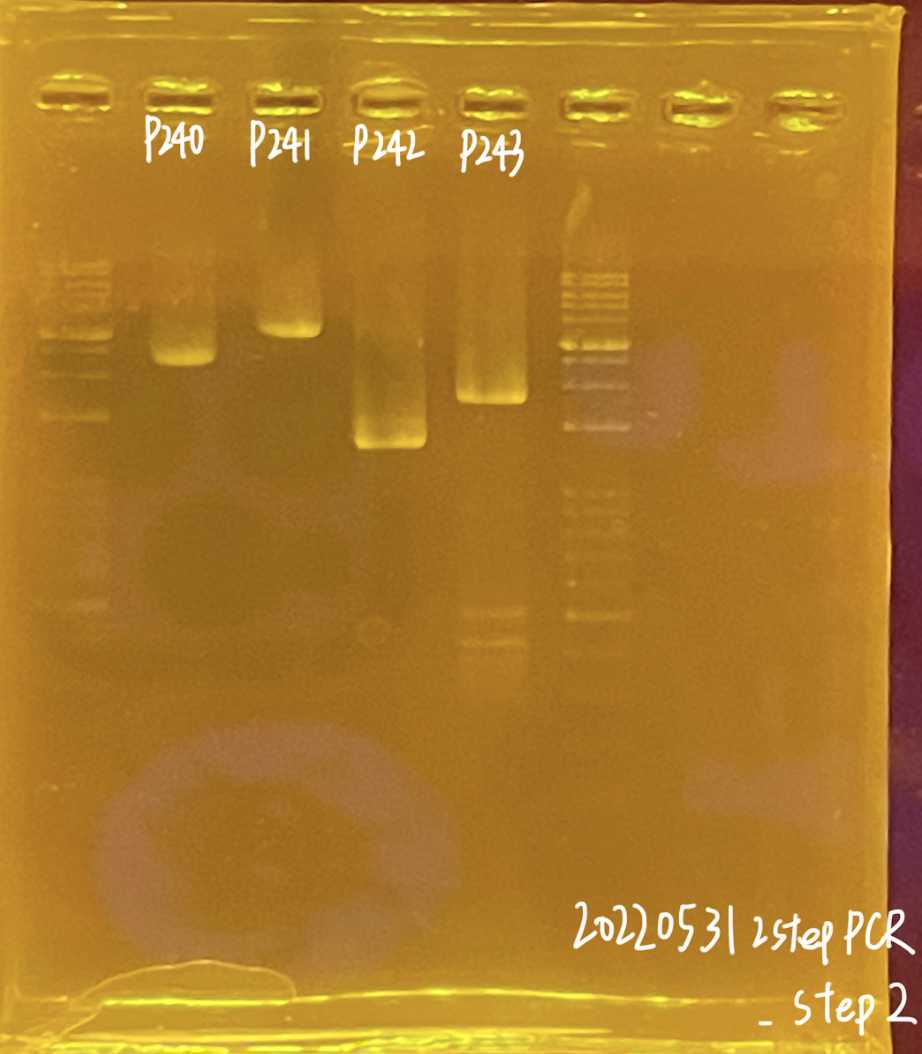
Marked gel picture go to the Lab Google drive
